Sam Brusco, Associate Editor03.05.24
BD (Becton Dickinson) has enrolled the first patient in its AGILITY investigational device exemption (IDE) study.
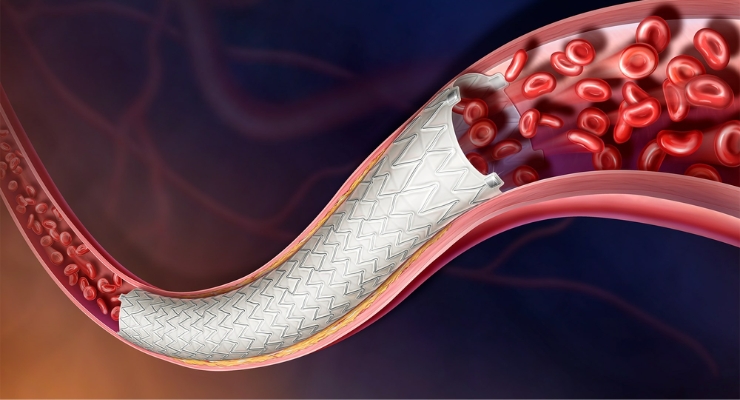
The AGILITY IDE study will assess the BD Vascular Covered Stent to treat peripheral arterial disease (PAD). The investigational stent is a self-expanding, low-profile, polytetrafluoroethylene (PTFE) encapsulated nitinol implant. It’s deployed via a delivery system that offers controlled stent release.
The clinical study will enroll 315 patients at up to 40 sites across the U.S., Europe, Australia, and New Zealand. Follow-up will begin at one month and end at 36 months.
Dr. Sean Lyden, chairman of the department of vascular surgery at Cleveland Clinic and national principal investigator of the AGILITY study said a self-expanding stent can play a crucial role to address advanced PAD.
"We need a stent that can track to the lesion, apposes the vessel wall and ultimately provides long-term durability,” Dr. Lyden told the press. “We're excited to see how this technology performs.”
Development of PAD includes formation of atherosclerosis and blood clots in leg arteries. Minimally invasive techniques with devices such as angioplasty balloons, drug-coated balloons, atherectomy, and covered stents can be used to increase blood flow through the diseased areas.
"There continues to be significant unmet needs in the treatment of PAD patients," said Tim Hug, VP and GM at BD Peripheral Intervention. "We are excited to have initiated this study and evaluate the treatment benefits of this potentially differentiated technology. This stent could give interventionalists an important new solution in the fight against PAD, expand our portfolio and enable us to better serve our customers and the patients they treat."
The AGILITY IDE study will assess the BD Vascular Covered Stent to treat peripheral arterial disease (PAD). The investigational stent is a self-expanding, low-profile, polytetrafluoroethylene (PTFE) encapsulated nitinol implant. It’s deployed via a delivery system that offers controlled stent release.
The clinical study will enroll 315 patients at up to 40 sites across the U.S., Europe, Australia, and New Zealand. Follow-up will begin at one month and end at 36 months.
Dr. Sean Lyden, chairman of the department of vascular surgery at Cleveland Clinic and national principal investigator of the AGILITY study said a self-expanding stent can play a crucial role to address advanced PAD.
"We need a stent that can track to the lesion, apposes the vessel wall and ultimately provides long-term durability,” Dr. Lyden told the press. “We're excited to see how this technology performs.”
Development of PAD includes formation of atherosclerosis and blood clots in leg arteries. Minimally invasive techniques with devices such as angioplasty balloons, drug-coated balloons, atherectomy, and covered stents can be used to increase blood flow through the diseased areas.
"There continues to be significant unmet needs in the treatment of PAD patients," said Tim Hug, VP and GM at BD Peripheral Intervention. "We are excited to have initiated this study and evaluate the treatment benefits of this potentially differentiated technology. This stent could give interventionalists an important new solution in the fight against PAD, expand our portfolio and enable us to better serve our customers and the patients they treat."