Sam Brusco, Associate Editor02.27.24
Fujifilm Healthcare Americas has been granted U.S. Food and Drug Administration (FDA) 510(k) clearance for Scale Eye, a new endoscopic imaging technology and part of the company’s growing Eluxeo endoscopic imaging system.
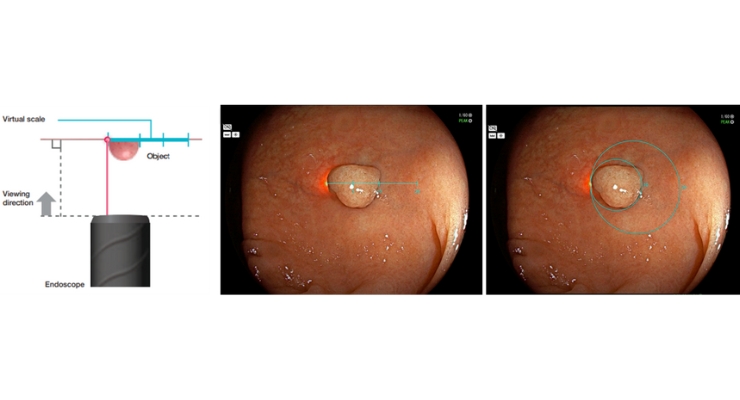
Scale Eye consists of a laser-equipped colonoscope and endoscopy support software and displays a linear or circular virtual measurement over the area of interest on the endoscopy monitor. Using it, endoscopists can estimate the size of colorectal lesions in vivo without needing visual estimations, consumable tools, or further surgical instruments.
The company said compared to a biopsy forceps methods, Scale Eye allows more accurate, objective colon polyp size measurement, an important factor for clinical management of neoplasms.
The company also shared data regarding Scale Eye’s use versus current methods in use. Its accuracy rate was 98.2% compared to 89% measuring with forceps, and measurement time was 2.8 seconds compared to eight seconds. Fujifilm also noted visual measurement overestimated polyp size in 57.3% of cases.
The company said it plans to commercially launch Scale Eye in 2024 following limited market evaluation.
“During colonoscopy, it is important to correctly measure the size of the polyps because it can directly impact the patient’s care pathway,” said Tai Fujita, VP, Endoscopy Division, Fujifilm Healthcare Americas Corporation. “Early clinical results of SCALE EYE are impressive, and we’re proud that Fujifilm’s new, innovative in vivo scaling capability is demonstrating success in improving both the speed and accuracy of polyp measurement.”
Scale Eye consists of a laser-equipped colonoscope and endoscopy support software and displays a linear or circular virtual measurement over the area of interest on the endoscopy monitor. Using it, endoscopists can estimate the size of colorectal lesions in vivo without needing visual estimations, consumable tools, or further surgical instruments.
The company said compared to a biopsy forceps methods, Scale Eye allows more accurate, objective colon polyp size measurement, an important factor for clinical management of neoplasms.
The company also shared data regarding Scale Eye’s use versus current methods in use. Its accuracy rate was 98.2% compared to 89% measuring with forceps, and measurement time was 2.8 seconds compared to eight seconds. Fujifilm also noted visual measurement overestimated polyp size in 57.3% of cases.
The company said it plans to commercially launch Scale Eye in 2024 following limited market evaluation.
“During colonoscopy, it is important to correctly measure the size of the polyps because it can directly impact the patient’s care pathway,” said Tai Fujita, VP, Endoscopy Division, Fujifilm Healthcare Americas Corporation. “Early clinical results of SCALE EYE are impressive, and we’re proud that Fujifilm’s new, innovative in vivo scaling capability is demonstrating success in improving both the speed and accuracy of polyp measurement.”