Sam Brusco, Associate Editor02.13.24
Tandem Diabetes Care has launched its new Tandem Mobi automated insulin delivery (AID) system in the U.S. According to the company, it’s the world’s smallest, durable AID system for diabetes patients.
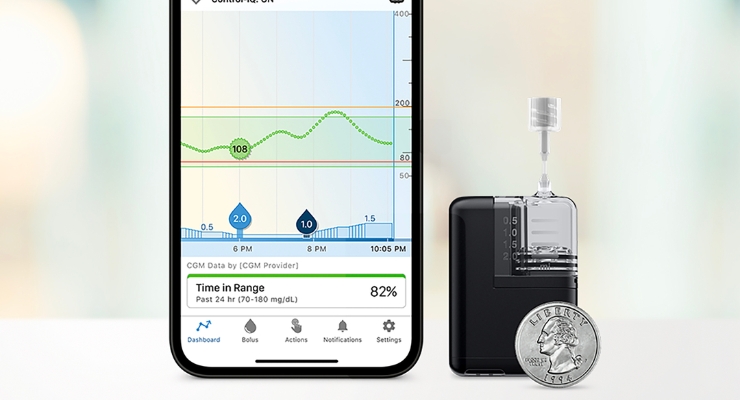
Less than half the size of the company’s t:slim X2 insulin pump, Tandem Mobi features the company’s Control-IQ technology, a hybrid closed-loop AID feature. It’s cleared for people with type 1 diabetes aged six and up. Control-IQ predicts and helps prevent high and low blood sugar, and has shown improved time in range throughout the day and night in clinical trials and real-world studies, according to the company.
The new AID received U.S. Food and Drug Administration (FDA) clearance in July 2023.
Tandem Mobi can be worn on-body with a lightweight adhesive sleeve, clipped onto clothing, or slipped into a pocket. It’s water resistant at a depth up to eight feet and for up to two hours.
Mobi is compatible with existing Tandem-branded infusion sets made by Convatec, including a new five-inch tubing option made specifically for Mobi. It’s compatible with the Dexcom G6 continuous glucose monitor (CGM) and the company said further CGM sensor integrations are planned, including Dexcom G7 in Q2 2024 and Abbott’s FreeStyle Libre 3 sensor later on.
Users have full iOS mobile control of Mobi with a compatible iPhone. Mobi also has a physical button to deliver insulin without their iPhone. It uses wireless charging and can receive remote software updates as needed.
“This tiny, wearable pump exceeded the expectations of users from an early access program, and we are thrilled to begin offering this exciting new technology to more people in the diabetes community,” said John Sheridan, Tandem’s president and CEO. "With this launch, we are executing on our strategy to offer a differentiated portfolio of durable insulin pumps, providing choice, along with new options in wearability.”
Less than half the size of the company’s t:slim X2 insulin pump, Tandem Mobi features the company’s Control-IQ technology, a hybrid closed-loop AID feature. It’s cleared for people with type 1 diabetes aged six and up. Control-IQ predicts and helps prevent high and low blood sugar, and has shown improved time in range throughout the day and night in clinical trials and real-world studies, according to the company.
The new AID received U.S. Food and Drug Administration (FDA) clearance in July 2023.
Tandem Mobi can be worn on-body with a lightweight adhesive sleeve, clipped onto clothing, or slipped into a pocket. It’s water resistant at a depth up to eight feet and for up to two hours.
Mobi is compatible with existing Tandem-branded infusion sets made by Convatec, including a new five-inch tubing option made specifically for Mobi. It’s compatible with the Dexcom G6 continuous glucose monitor (CGM) and the company said further CGM sensor integrations are planned, including Dexcom G7 in Q2 2024 and Abbott’s FreeStyle Libre 3 sensor later on.
Users have full iOS mobile control of Mobi with a compatible iPhone. Mobi also has a physical button to deliver insulin without their iPhone. It uses wireless charging and can receive remote software updates as needed.
“This tiny, wearable pump exceeded the expectations of users from an early access program, and we are thrilled to begin offering this exciting new technology to more people in the diabetes community,” said John Sheridan, Tandem’s president and CEO. "With this launch, we are executing on our strategy to offer a differentiated portfolio of durable insulin pumps, providing choice, along with new options in wearability.”