Sam Brusco, Associate Editor02.13.24
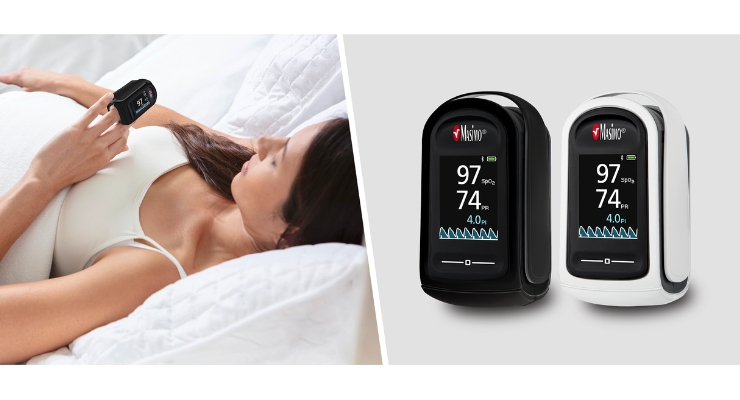
Masimo has received U.S. Food and Drug Administration (FDA) clearance for MightySat Medical, a medical fingertip pulse oximeter that’s available over-the-counter (OTC) direct to consumers, without a prescription. It’s the first such device of its kind, according to the company.
MightySat Medical is meant for spot-checking of functional oxygen saturation of arterial hemoglobin (SpO2) and pulse rate. It offers accurate, reliable measurements on patients of all skin colors and patients with low perfusion.
It provides a direct-to-consumer option for medical device with hospital-grade pulse oximetry technology, Masimo SET. The company said that over 100 independent and objective studies has demonstrated that SET outperforms other pulse oximetry technologies.
“Until now, consumers and even healthcare providers had no way of knowing what pulse oximeter they could trust to use at home,” said Masimo founder and CEO Joe Kiani. “On the internet and even in drug stores, they are inundated with a myriad of products that are unreliable, with misleading advertisements about their abilities to provide accurate measurements of oxygen saturation and pulse rate.”
“This clearance of MightySat Medical for consumers eliminates the confusion, placing an FDA-cleared, accurate, reliable, and revolutionary SET pulse oximeter, with technology that hospitals have been using for more than 25 years, directly into their hands,” Kiani continued. “Healthcare providers can also now be confident when referring their patients to get MightySat Medical knowing that it has actually been cleared by the FDA as an OTC medical pulse oximeter.”
In December 2023, Masimo earned FDA clearance of its Stork baby monitoring system for prescription, home use for healthy and sick babies 0-18 months old.
MightySat Medical is meant for spot-checking of functional oxygen saturation of arterial hemoglobin (SpO2) and pulse rate. It offers accurate, reliable measurements on patients of all skin colors and patients with low perfusion.
It provides a direct-to-consumer option for medical device with hospital-grade pulse oximetry technology, Masimo SET. The company said that over 100 independent and objective studies has demonstrated that SET outperforms other pulse oximetry technologies.
“Until now, consumers and even healthcare providers had no way of knowing what pulse oximeter they could trust to use at home,” said Masimo founder and CEO Joe Kiani. “On the internet and even in drug stores, they are inundated with a myriad of products that are unreliable, with misleading advertisements about their abilities to provide accurate measurements of oxygen saturation and pulse rate.”
“This clearance of MightySat Medical for consumers eliminates the confusion, placing an FDA-cleared, accurate, reliable, and revolutionary SET pulse oximeter, with technology that hospitals have been using for more than 25 years, directly into their hands,” Kiani continued. “Healthcare providers can also now be confident when referring their patients to get MightySat Medical knowing that it has actually been cleared by the FDA as an OTC medical pulse oximeter.”
In December 2023, Masimo earned FDA clearance of its Stork baby monitoring system for prescription, home use for healthy and sick babies 0-18 months old.