Sam Brusco, Associate Editor02.08.24
Aerin Medical has published two-year outcomes from its VATRAC trial in Rhinology Journal for its VivAer treatment of nasal valve collapse.
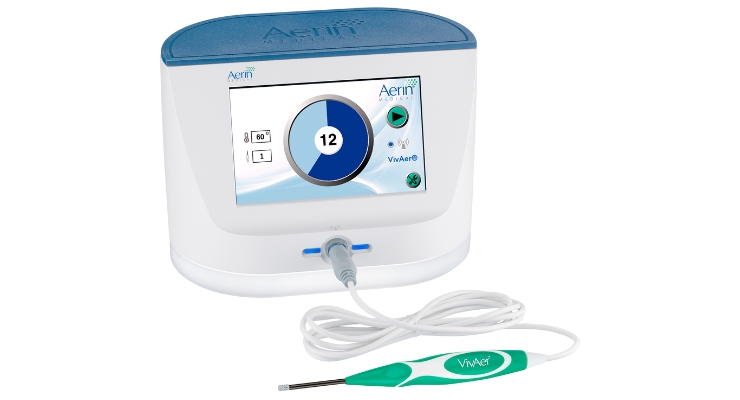
VivAer offers ENT physicians a non-invasive option that durably remodels tissue in the nasal valve to address nasal airway obstruction (NAO) in appropriate patients. It features a thin, wand-like stylus that attaches to a console. The stylus is inserted via the nostril to gently remodel nasal tissue and improve airflow. It doesn’t involve any cutting, removal of nasal tissue or bone, or use of an implant.
The study demonstrated, according to the company, that a single in-office treatment with its VivAer resulted in sustained improvement in NAO symptoms caused by nasal valve collapse. It also showed reduced daytime sleepiness and use of medications and mechanical aids.
The VATRAC trial enrolled 119 patients with extreme or severe NAO based on the Nasal Obstruction Symptom Evaluation (NOSE) Scale score, with nasal valve collapse as a primary or significant contributor to symptoms. After analysis of the primary trial endpoint, 31 control patients eligible for crossover elected to undergo VivAer treatment.
Patients reported “durable improvements from baseline” though 24 months after the procedure. Findings included:
“This publication adds to the deep dossier of clinical evidence for VivAer, with three separate studies now confirming multi-year benefit and with comparable responder rates,” said Matt Brokaw, CEO of Aerin. “We extend our gratitude to the clinical investigators whose dedication has been instrumental in demonstrating that VivAer is a practical and effective innovation changing how and where nasal airway obstruction patients are treated.”
In September 2023, the company shared positive two-year follow-up data from its AERWAY trial.
VivAer offers ENT physicians a non-invasive option that durably remodels tissue in the nasal valve to address nasal airway obstruction (NAO) in appropriate patients. It features a thin, wand-like stylus that attaches to a console. The stylus is inserted via the nostril to gently remodel nasal tissue and improve airflow. It doesn’t involve any cutting, removal of nasal tissue or bone, or use of an implant.
The study demonstrated, according to the company, that a single in-office treatment with its VivAer resulted in sustained improvement in NAO symptoms caused by nasal valve collapse. It also showed reduced daytime sleepiness and use of medications and mechanical aids.
The VATRAC trial enrolled 119 patients with extreme or severe NAO based on the Nasal Obstruction Symptom Evaluation (NOSE) Scale score, with nasal valve collapse as a primary or significant contributor to symptoms. After analysis of the primary trial endpoint, 31 control patients eligible for crossover elected to undergo VivAer treatment.
Patients reported “durable improvements from baseline” though 24 months after the procedure. Findings included:
- Responder rate at two years was 90.4%, sustained across all time periods
- At two years post-treatment, patients maintained a 54.7% improvement in NOSE scores compared to baseline
- Presence of septal deviation didn’t significantly affect treatment outcomes
- Treatment with VivAer was well-tolerated; no serious adverse events related to the procedure or device
“This publication adds to the deep dossier of clinical evidence for VivAer, with three separate studies now confirming multi-year benefit and with comparable responder rates,” said Matt Brokaw, CEO of Aerin. “We extend our gratitude to the clinical investigators whose dedication has been instrumental in demonstrating that VivAer is a practical and effective innovation changing how and where nasal airway obstruction patients are treated.”
In September 2023, the company shared positive two-year follow-up data from its AERWAY trial.