Rachel Klemovitch, Assistant Editor02.01.24
USGI Medical, a company specializing in incisionless weight loss procedures and devices, announced that the U.S. Food and Drug Administration (FDA) has approved an Investigational Device Exemption (IDE) to conduct a study of its POSE2.0 incisionless procedure for primary obesity.
The clinical trial INSPIRO (incisionless Suture Plications (POSE2.0) Ina Randomized Obesity Study) will evaluate up to 186 participants in seven sites across the U.S. and possible participants outside the U.S.
Interim CEO of USGI Medical, Arnold Podgorsky told the press, “The FDA’s action is another important milestone for USGI, and we are excited to move forward with a pivotal study to further validate POSE2.0’s safety, durability and effectiveness in treating primary obesity. We appreciate the FDA’s support as we look to advance this potentially life-changing treatment option in the United States, which has already helped thousands of patients in other countries.”
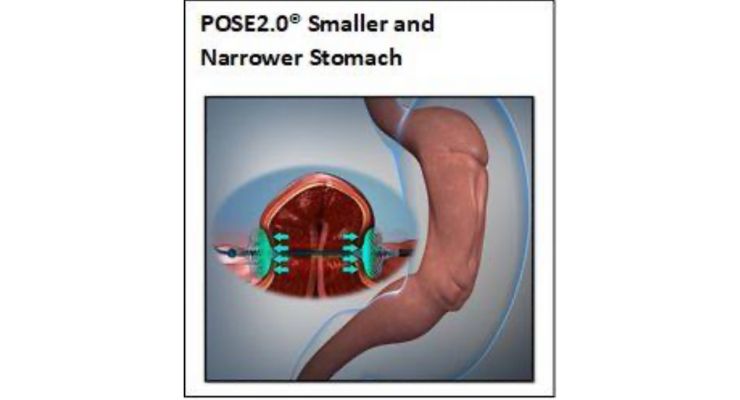
POSE2.0 utilizes the company’s Incisionless Operating Platform (IOP) to place Snowshoe Suture Anchors in gastric tissue to narrow and shorten the stomach. Similar to the results of a surgical sleeve gastrectomy, POSE2.0 provides volumetric gastric restriction without surgically removing part of the stomach.
According to USGI, endoscopic gastric remodeling is an emerging approach to weight loss procedures that does not require the removal of the stomach.
Doctor Barham Abu Dayyeh, at Mayo Clinic is expected to lead the INSPIRO study alongside Doctor Erik Wilson at the University of Texas McGovern Medical at Houston.
Dayyeh served as Principal Investigator USGI’s pilot study of POSE2.0 that included 40 patients suffering from obesity with an obesity-related comorbidity at four clinical centers.
“This procedure holds promise, as it could prove to be a cost-effective, less burdensome therapeutic option with value added over existing pharmaceutical and surgical approaches,” Podgorsky added. “Marketing in the U.S. would be a game-changer for USGI, its investors and the doctor-patient community.”
The clinical trial INSPIRO (incisionless Suture Plications (POSE2.0) Ina Randomized Obesity Study) will evaluate up to 186 participants in seven sites across the U.S. and possible participants outside the U.S.
Interim CEO of USGI Medical, Arnold Podgorsky told the press, “The FDA’s action is another important milestone for USGI, and we are excited to move forward with a pivotal study to further validate POSE2.0’s safety, durability and effectiveness in treating primary obesity. We appreciate the FDA’s support as we look to advance this potentially life-changing treatment option in the United States, which has already helped thousands of patients in other countries.”
POSE2.0 utilizes the company’s Incisionless Operating Platform (IOP) to place Snowshoe Suture Anchors in gastric tissue to narrow and shorten the stomach. Similar to the results of a surgical sleeve gastrectomy, POSE2.0 provides volumetric gastric restriction without surgically removing part of the stomach.
According to USGI, endoscopic gastric remodeling is an emerging approach to weight loss procedures that does not require the removal of the stomach.
Doctor Barham Abu Dayyeh, at Mayo Clinic is expected to lead the INSPIRO study alongside Doctor Erik Wilson at the University of Texas McGovern Medical at Houston.
Dayyeh served as Principal Investigator USGI’s pilot study of POSE2.0 that included 40 patients suffering from obesity with an obesity-related comorbidity at four clinical centers.
“This procedure holds promise, as it could prove to be a cost-effective, less burdensome therapeutic option with value added over existing pharmaceutical and surgical approaches,” Podgorsky added. “Marketing in the U.S. would be a game-changer for USGI, its investors and the doctor-patient community.”