Michael Barbella, Managing Editor01.26.24
Hyperfine Inc. has announced initial findings from a multicenter observational study Assessing Brain Injury Using Portable Magnetic Resonance Imaging in Patients With Extracorporeal Membrane Oxygenation.
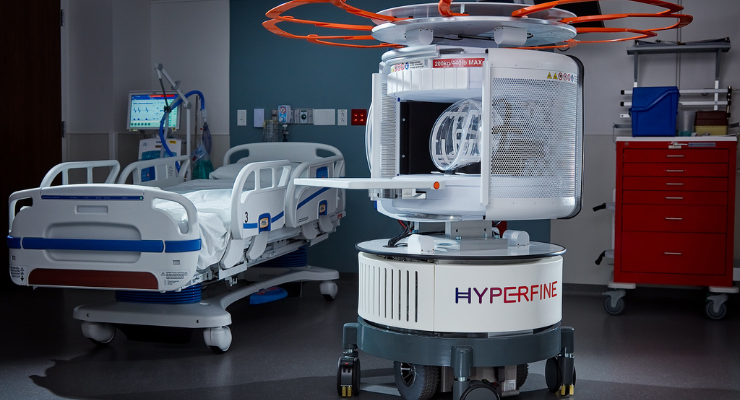
The study evaluated the efficacy of the Swoop system, touted as the world's first U.S. Food and Drug Administration (FDA)-cleared portable, ultra-low-field magnetic resonance (MR) brain imaging system. Clinicians used Swoop to assess acute brain injury (ABI) in 46 adult extracorporeal membrane oxygenation (ECMO) patients at two major U.S. medical institutions. ECMO patients are typically unable to be transported to the conventional high-field magnetic resonance imaging suite due to their unstable condition. The study evaluated the feasibility of using portable MR brain imaging for monitoring patients during ECMO use, highlighting its potential advantage in providing early visualization of ischemic stroke compared to computerized tomography (CT) scans.
“The study’s findings are very encouraging, especially as they relate to the benefits of using the Swoop portable brain imaging system for early assessment of acute brain injury in patients being treated with ECMO. Without the Swoop system, these patients, who are too unstable to transport, would not have access to the benefits of MR brain imaging,” Hyperfine President/CEO Maria Sainz said. “This data further supports yet another use case for the Swoop system where timely access to MR brain imaging for patients and their clinical teams can make a profound impact on mitigating care delays.”
Initial trial findings demonstrate the feasibility of using portable MR brain imaging with adult ECMO patients. It concludes that the incidence of acute brain injury was high, with ischemic stroke being the most common type of injury. The study further underscores the role of MR-based neuroimaging in ABI detection and the potential for allowing improvement in neurological outcomes.
“This study signals the potential benefit of portable MR brain imaging over conventional CT scans during ECMO, especially in detecting early ischemic injuries,” Hyperfine Vice President of Medical Affairs Edmond A. Knopp, M.D., stated. “This may be particularly important when you consider mortality rates due to acute brain injury in ECMO patients and emphasizes the need for early and accurate neuromonitoring.”
Hyperfine Inc. is redefining brain imaging with the Swoop system, which received initial FDA clearance in 2020 as a portable magnetic resonance brain imaging device for producing images that display the head's internal structure when a full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. The Swoop system has been approved for brain imaging in several countries, including Canada and Australia, has UKCA certification in the United Kingdom, CE certification in the European Union, and is also available in New Zealand.
Founded by Dr. Jonathan Rothberg in a technology-based incubator called 4Catalyzer, Hyperfine scientists, engineers, and physicists developed the Swoop system out of a passion for redefining brain imaging methodology and the ways in which clinicians apply accessible diagnostic imaging to patient care. Traditionally, access to costly, stationary, conventional MRI technology can be inconvenient or not available when needed most. With the portable, ultra-low-field Swoop system, Hyperfine is redefining the neuroimaging workflow by bringing brain imaging to the patient’s bedside.
Hyperfine, Swoop, and Portable MR Imaging are registered trademarks of Hyperfine Inc.
The study evaluated the efficacy of the Swoop system, touted as the world's first U.S. Food and Drug Administration (FDA)-cleared portable, ultra-low-field magnetic resonance (MR) brain imaging system. Clinicians used Swoop to assess acute brain injury (ABI) in 46 adult extracorporeal membrane oxygenation (ECMO) patients at two major U.S. medical institutions. ECMO patients are typically unable to be transported to the conventional high-field magnetic resonance imaging suite due to their unstable condition. The study evaluated the feasibility of using portable MR brain imaging for monitoring patients during ECMO use, highlighting its potential advantage in providing early visualization of ischemic stroke compared to computerized tomography (CT) scans.
“The study’s findings are very encouraging, especially as they relate to the benefits of using the Swoop portable brain imaging system for early assessment of acute brain injury in patients being treated with ECMO. Without the Swoop system, these patients, who are too unstable to transport, would not have access to the benefits of MR brain imaging,” Hyperfine President/CEO Maria Sainz said. “This data further supports yet another use case for the Swoop system where timely access to MR brain imaging for patients and their clinical teams can make a profound impact on mitigating care delays.”
Initial trial findings demonstrate the feasibility of using portable MR brain imaging with adult ECMO patients. It concludes that the incidence of acute brain injury was high, with ischemic stroke being the most common type of injury. The study further underscores the role of MR-based neuroimaging in ABI detection and the potential for allowing improvement in neurological outcomes.
“This study signals the potential benefit of portable MR brain imaging over conventional CT scans during ECMO, especially in detecting early ischemic injuries,” Hyperfine Vice President of Medical Affairs Edmond A. Knopp, M.D., stated. “This may be particularly important when you consider mortality rates due to acute brain injury in ECMO patients and emphasizes the need for early and accurate neuromonitoring.”
Hyperfine Inc. is redefining brain imaging with the Swoop system, which received initial FDA clearance in 2020 as a portable magnetic resonance brain imaging device for producing images that display the head's internal structure when a full diagnostic examination is not clinically practical. When interpreted by a trained physician, these images provide information that can be useful in determining a diagnosis. The Swoop system has been approved for brain imaging in several countries, including Canada and Australia, has UKCA certification in the United Kingdom, CE certification in the European Union, and is also available in New Zealand.
Founded by Dr. Jonathan Rothberg in a technology-based incubator called 4Catalyzer, Hyperfine scientists, engineers, and physicists developed the Swoop system out of a passion for redefining brain imaging methodology and the ways in which clinicians apply accessible diagnostic imaging to patient care. Traditionally, access to costly, stationary, conventional MRI technology can be inconvenient or not available when needed most. With the portable, ultra-low-field Swoop system, Hyperfine is redefining the neuroimaging workflow by bringing brain imaging to the patient’s bedside.
Hyperfine, Swoop, and Portable MR Imaging are registered trademarks of Hyperfine Inc.