Michael Barbella, Managing Editor01.08.24
Linear Health Sciences has received an expanded indication from the U.S. Food and Drug Administration (FDA) for its Orchid SRV safety release valve. The expanded indication allows the Orchid SRV to be used with all IV access methods.
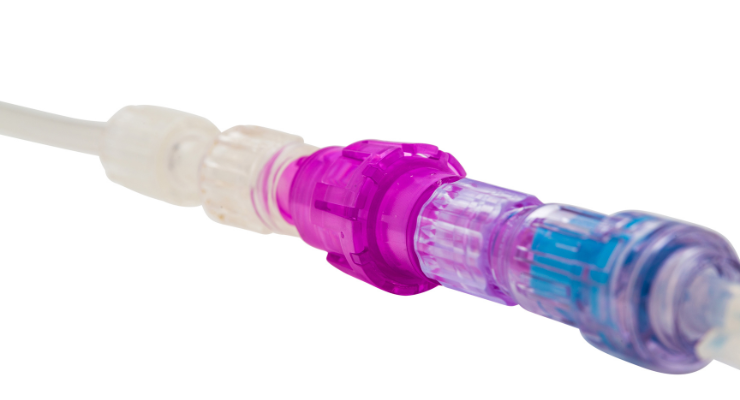
The Orchid SRV is a sterile, single-use connector for needle-free access that, when activated, makes return to treatment fast, simple, and clean, while improving both the patient and clinician experience. It is now indicated for use with central venous catheters, peripherally inserted central catheters (PICCs), peripheral IV catheters, and intraosseous cannulation, during direct injection, intermittent infusion, and continuous infusion, in patients two weeks of age and older.
“Maintaining vascular access is one of the most fundamental, yet most over-looked, treatment modalities in medicine today,” Linear Health Sciences Co-Founder/Chief Medical Officer Ryan Dennis said. “Expanding indication for the Orchid SRV to all IV settings will help improve outcomes for millions of patients every year.”
Dislodgement and associated IV treatment failures in the more than 342 million peripheral IVs administered in the United States each year1 are estimated to cost the American healthcare system more than $2 billion annually.2 Dislodgement is most frequently caused by patient confusion or removal of the catheter, loose dressing or tape, or tubing becoming tangled in bed linens. Clinical simulation testing of 360 Orchid SRVs showed that the device prevented IV dislodgement by 91.1% across all test groups.
“The expanded indication from the FDA represents an acceleration of our commercialization efforts, as we look to create a continuum of care for vascular access,” Linear Health Sciences Co-Founder/CEO Dan Clark stated. “In addition to improved care for patients, the Orchid SRV enables significant downstream cost savings, which benefits the entire healthcare ecosystem.”
Linear Health Sciences won an Innovative Technology Award for Orchid SRV from Vizient, making the device available to hospital customers through a non-exclusive contract with the group purchasing organization (GPO). Additionally, the device is available nationwide and across Canada. Qualified clinical evaluations of the Orchid SRV will continually quantify device utilization.
Linear Health Sciences has developed a proprietary, breakaway safety valve technology to help improve the use of medical tubing in hospitals. The platform technology was developed to increase the safety and satisfaction of patients, caregivers, and healthcare facilities, while dramatically reducing costs. The company’s initial products include the Orchid SRV for use in IV catheter therapy and the Orchid SRV Type D device for use in surgical/wound, nephrostomy and abscess drainage.
References
1 iData research report
2 "The Peripheral Intravenous Catheter Journey: A Prospective Cohort Study of 1000 Patients." Podium presentation by Nicole Marsh, RN, and Claire Rickard, RN, PhD, AVA 2017 annual meeting.
The Orchid SRV is a sterile, single-use connector for needle-free access that, when activated, makes return to treatment fast, simple, and clean, while improving both the patient and clinician experience. It is now indicated for use with central venous catheters, peripherally inserted central catheters (PICCs), peripheral IV catheters, and intraosseous cannulation, during direct injection, intermittent infusion, and continuous infusion, in patients two weeks of age and older.
“Maintaining vascular access is one of the most fundamental, yet most over-looked, treatment modalities in medicine today,” Linear Health Sciences Co-Founder/Chief Medical Officer Ryan Dennis said. “Expanding indication for the Orchid SRV to all IV settings will help improve outcomes for millions of patients every year.”
Dislodgement and associated IV treatment failures in the more than 342 million peripheral IVs administered in the United States each year1 are estimated to cost the American healthcare system more than $2 billion annually.2 Dislodgement is most frequently caused by patient confusion or removal of the catheter, loose dressing or tape, or tubing becoming tangled in bed linens. Clinical simulation testing of 360 Orchid SRVs showed that the device prevented IV dislodgement by 91.1% across all test groups.
“The expanded indication from the FDA represents an acceleration of our commercialization efforts, as we look to create a continuum of care for vascular access,” Linear Health Sciences Co-Founder/CEO Dan Clark stated. “In addition to improved care for patients, the Orchid SRV enables significant downstream cost savings, which benefits the entire healthcare ecosystem.”
Linear Health Sciences won an Innovative Technology Award for Orchid SRV from Vizient, making the device available to hospital customers through a non-exclusive contract with the group purchasing organization (GPO). Additionally, the device is available nationwide and across Canada. Qualified clinical evaluations of the Orchid SRV will continually quantify device utilization.
Linear Health Sciences has developed a proprietary, breakaway safety valve technology to help improve the use of medical tubing in hospitals. The platform technology was developed to increase the safety and satisfaction of patients, caregivers, and healthcare facilities, while dramatically reducing costs. The company’s initial products include the Orchid SRV for use in IV catheter therapy and the Orchid SRV Type D device for use in surgical/wound, nephrostomy and abscess drainage.
References
1 iData research report
2 "The Peripheral Intravenous Catheter Journey: A Prospective Cohort Study of 1000 Patients." Podium presentation by Nicole Marsh, RN, and Claire Rickard, RN, PhD, AVA 2017 annual meeting.