Sam Brusco, Associate Editor01.05.24
Nanowear, a company focused on home, remote diagnostics, has gained the fourth U.S. Food and Drug Administration (FDA) 510(k) clearance for its SimpleSense nanotechnology-enabled wearable platform.
The clearance covers SimpleSense-BP, a novel artificial intelligence (AI)-based Software-as-a-Medical Device (SaMD)
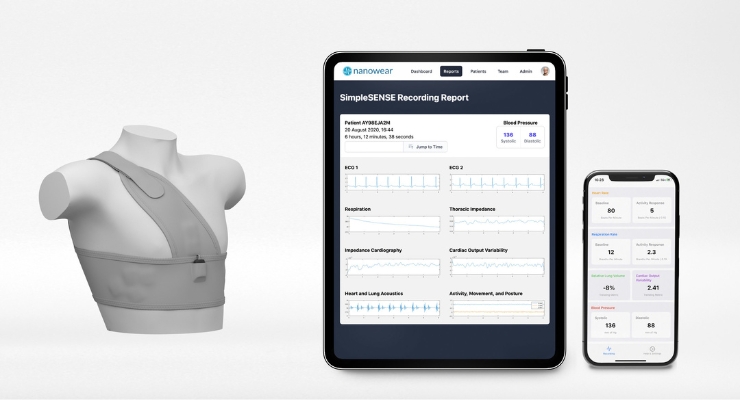
The system consists of a non-invasive, cuffless, continuous blood pressure monitor and the SimpleSense BP diagnostic. The company said it could be a transformational tool for clinical diagnostic management of high blood pressure and adds to the platform’s previously cleared cardiopulmonary diagnostics.
According to Nanowear, SimpleSense captures and analyzes 85+ unique biomarker data points across the heart, lungs, hemodynamic, and central vascular system. The platform aggregates millisecond by millisecond cardiopulmonary assessments to help medical professionals with remote patient management. Using proprietary nanosensors and algorithms, SimpleSense could replace blood pressure lines, digital stethoscopes, multi-channel ECGs, hemodynamic monitors, pulse oximeters, spirometers, and actigraphy monitors.
SimpleSense is indicated for use at home, in healthcare facilities, and clinical research. Its SimpleSense BP 4-decision tree algorithmic model was tested in three study arms and is validated to track changes of > +/- 15mmHg systolic and +/- 10mmHg diastolic blood pressures over a continuous recording period across all hypertension classes via its wearable undergarment and artificial intelligence (AI) platform.
“This FDA clearance represents an exciting and differentiating moment for Nanowear. Accelerated by the pandemic, the market has been eagerly searching for reliable, clinical-grade wearable, and data service solutions that find product market fit with the following three attributes: at-home, utilizing AI, and a proven team that executes,” said Venk Varadan, CEO and co-founder of Nanowear. “This SaMD is just the first example of what our nanotechnology and AI is capable of, in understanding the individualized risk signatures and cardiopulmonary care pathways of each patient, materially enabling precision medicine.”
The clearance covers SimpleSense-BP, a novel artificial intelligence (AI)-based Software-as-a-Medical Device (SaMD)
The system consists of a non-invasive, cuffless, continuous blood pressure monitor and the SimpleSense BP diagnostic. The company said it could be a transformational tool for clinical diagnostic management of high blood pressure and adds to the platform’s previously cleared cardiopulmonary diagnostics.
According to Nanowear, SimpleSense captures and analyzes 85+ unique biomarker data points across the heart, lungs, hemodynamic, and central vascular system. The platform aggregates millisecond by millisecond cardiopulmonary assessments to help medical professionals with remote patient management. Using proprietary nanosensors and algorithms, SimpleSense could replace blood pressure lines, digital stethoscopes, multi-channel ECGs, hemodynamic monitors, pulse oximeters, spirometers, and actigraphy monitors.
SimpleSense is indicated for use at home, in healthcare facilities, and clinical research. Its SimpleSense BP 4-decision tree algorithmic model was tested in three study arms and is validated to track changes of > +/- 15mmHg systolic and +/- 10mmHg diastolic blood pressures over a continuous recording period across all hypertension classes via its wearable undergarment and artificial intelligence (AI) platform.
“This FDA clearance represents an exciting and differentiating moment for Nanowear. Accelerated by the pandemic, the market has been eagerly searching for reliable, clinical-grade wearable, and data service solutions that find product market fit with the following three attributes: at-home, utilizing AI, and a proven team that executes,” said Venk Varadan, CEO and co-founder of Nanowear. “This SaMD is just the first example of what our nanotechnology and AI is capable of, in understanding the individualized risk signatures and cardiopulmonary care pathways of each patient, materially enabling precision medicine.”