Sam Brusco, Associate Editor11.15.23
The U.S. Food and Drug Administration (FDA) has deemed Cardinal Health’s September 19th recall of its Monoject single-use Luer lock syringes (1, 6, 12, 20, 35, and 60mL) as Class I.
The Monoject disposable syringes inject fluid into or withdraw fluids from the body. When used with syringe pumps, they are loaded with fluid or medications and placed in the pump.
In June 2023, the company began distributing the syringes branded as “Cardinal Health Monoject syringes,” which differ from the previously branded “Covidien Monoject syringes” because they have different dimensions. The company advised that the affected Cardinal Health-branded Monoject syringes shouldn’t be used with syringe pumps.
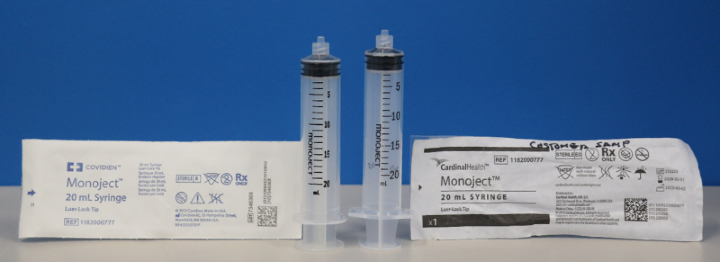
The Covidien Monoject syringe on the left has different dimensions than the Cardinal Health Monoject syringe on the right.
The company warned the size changes made to the affected syringes, when used in pumps, could cause overdose, underdose, therapy delay, or occlusion alarm delay. Thus far Cardinal has received 15 delayed therapy reports due to pumps not recognizing syringes, and 13 reports of inaccurate volume/rate dispensing—including some injuries.
No deaths have been reported as of yet. The FDA also urged to be aware that the recall is a correction, not a recall.
The Monoject disposable syringes inject fluid into or withdraw fluids from the body. When used with syringe pumps, they are loaded with fluid or medications and placed in the pump.
In June 2023, the company began distributing the syringes branded as “Cardinal Health Monoject syringes,” which differ from the previously branded “Covidien Monoject syringes” because they have different dimensions. The company advised that the affected Cardinal Health-branded Monoject syringes shouldn’t be used with syringe pumps.

The Covidien Monoject syringe on the left has different dimensions than the Cardinal Health Monoject syringe on the right.
The company warned the size changes made to the affected syringes, when used in pumps, could cause overdose, underdose, therapy delay, or occlusion alarm delay. Thus far Cardinal has received 15 delayed therapy reports due to pumps not recognizing syringes, and 13 reports of inaccurate volume/rate dispensing—including some injuries.
No deaths have been reported as of yet. The FDA also urged to be aware that the recall is a correction, not a recall.












