Sam Brusco, Associate Editor11.09.23
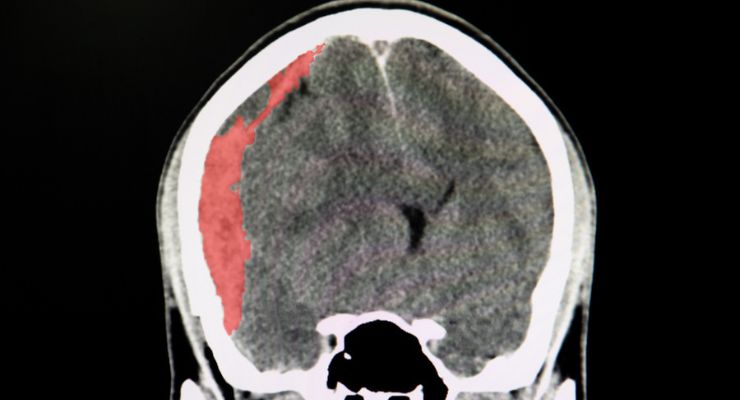
RapidAI has earned U.S. Food and Drug Administration (FDA) clearance for Rapid SDH its artificial intelligence (AI)-powered module to detect and notify of suspected hemispheric acute and chronic subdural hematoma (SDH).
Rapid SDH joins Rapid ICH and Hyperdensity in the company’s hemorrhagic and trauma care AI solution, seeks to help frontline healthcare professionals streamline decision-making and expedite care. It uses its AI and intelligence gathered from analysis of hundreds of patient scans to help neurocritical care teams spot suspected hemispheric subdural hemorrhage.
The accompanying mobile app automatically sends results to the trauma team, which the company said can be received in under a minute. RapidAI also pointed to a Positive Predictive Value (PPV) of 99% (sensitivity 93%, specificity 99%) for suspected acute and chronic hemispheric subdural hematomas >1mL.
"The FDA's clearance of Rapid SDH significantly enhances our expanding range of hemorrhagic and trauma care solutions at this crucial time of rapidly growing patient numbers, clinician shortages, and advancements in potential treatment options,” RapidAI’s chief technology officer Amit Phadnis told the press. “Our goal is to continue to expand the capabilities and applications of our deep AI to deliver comprehensive clinical solutions that provide care teams with the crucial insights necessary to evaluate patients, streamline decision making, and expedite care for this common and dangerous disease.”
Rapid SDH joins Rapid ICH and Hyperdensity in the company’s hemorrhagic and trauma care AI solution, seeks to help frontline healthcare professionals streamline decision-making and expedite care. It uses its AI and intelligence gathered from analysis of hundreds of patient scans to help neurocritical care teams spot suspected hemispheric subdural hemorrhage.
The accompanying mobile app automatically sends results to the trauma team, which the company said can be received in under a minute. RapidAI also pointed to a Positive Predictive Value (PPV) of 99% (sensitivity 93%, specificity 99%) for suspected acute and chronic hemispheric subdural hematomas >1mL.
"The FDA's clearance of Rapid SDH significantly enhances our expanding range of hemorrhagic and trauma care solutions at this crucial time of rapidly growing patient numbers, clinician shortages, and advancements in potential treatment options,” RapidAI’s chief technology officer Amit Phadnis told the press. “Our goal is to continue to expand the capabilities and applications of our deep AI to deliver comprehensive clinical solutions that provide care teams with the crucial insights necessary to evaluate patients, streamline decision making, and expedite care for this common and dangerous disease.”