Sam Brusco, Associate Editor11.01.23
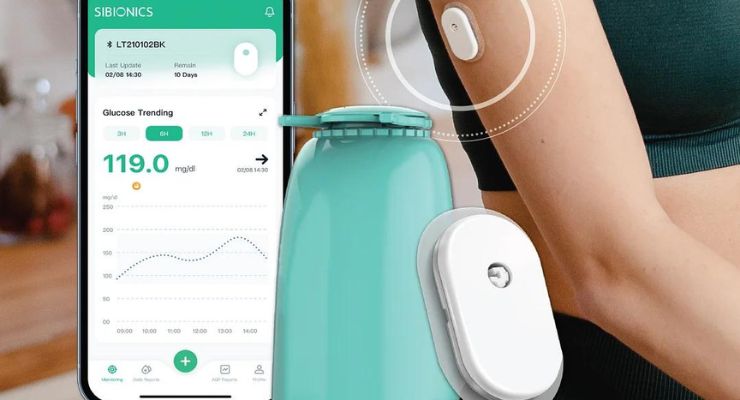
SIBIONICS, a continuous glucose monitoring (CGM) maker, has earned CE mark clearance for its GS1 CGM system.
SIBIONICS was established in 2015 and has a workforce of over 700 people, 40% of which the company said is actively engaged in R&D efforts. The company also said that as of today, it has collaborated with 1,600 hospitals to adopt the GS1 CGM for inpatient treatment and more than 600,000 users have managed their diabetes with GS1 worldwide.
The GS1 CGM features calibration-free, 14 days of continuous glucose monitoring. Glucose data can be transmitted to monitoring devices or mobile applications, and create professional AGP reports. It also supports sharing data with friends and family, and integrates the latest ProView remote access platform for timely interventions.
It received marketing approval in China in November 2021, becoming the first blood-glucose meter without the need for finger blood calibration, SIBIONICS claims.
The company has also launched its consumer-grade biowearable, the KS1 continuous ketone monitoring (CKM) system. The device helps patients achieve and maintain ketosis, when the body uses fat instead of glucose as its main energy source. It features up to 14 days of uninterrupted usage, real-time tracking via an app, and personalized ketogenic diet guidance.
SIBIONICS was established in 2015 and has a workforce of over 700 people, 40% of which the company said is actively engaged in R&D efforts. The company also said that as of today, it has collaborated with 1,600 hospitals to adopt the GS1 CGM for inpatient treatment and more than 600,000 users have managed their diabetes with GS1 worldwide.
The GS1 CGM features calibration-free, 14 days of continuous glucose monitoring. Glucose data can be transmitted to monitoring devices or mobile applications, and create professional AGP reports. It also supports sharing data with friends and family, and integrates the latest ProView remote access platform for timely interventions.
It received marketing approval in China in November 2021, becoming the first blood-glucose meter without the need for finger blood calibration, SIBIONICS claims.
The company has also launched its consumer-grade biowearable, the KS1 continuous ketone monitoring (CKM) system. The device helps patients achieve and maintain ketosis, when the body uses fat instead of glucose as its main energy source. It features up to 14 days of uninterrupted usage, real-time tracking via an app, and personalized ketogenic diet guidance.