Michael Barbella, Managing Editor10.31.23
Epineuron has enrolled the first patient in its REGAIN pivotal trial. The multicenter, randomized, sham-controlled, double-blinded study is evaluating the safety and effectiveness of PeriPulse, an investigational technology designated a U.S. Food and Drug Administration Breakthrough Device for treating injured peripheral nerves.
The study aims to enroll up to 110 patients across Canada and the United States, making it the largest medical device study for nerve regeneration.
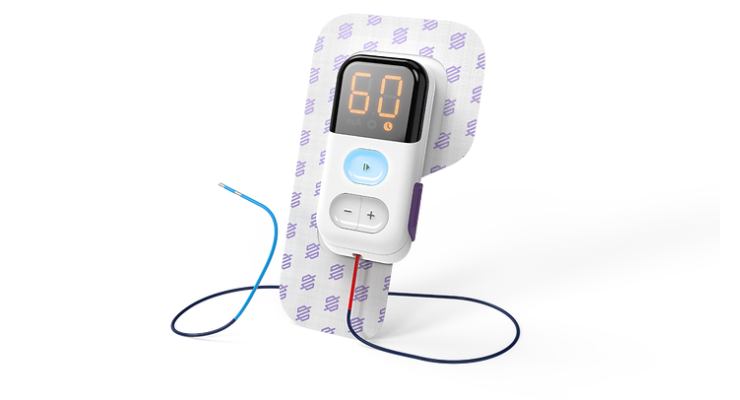
PeriPulse is a direct clinical translation of the research led by Drs. Tessa Gordon and Ming Chan from the University of Alberta spanning basic science to early clinical studies investigating the effects of bioelectronic therapy combined with standard nerve interventions. The proprietary system has been engineered to enhance usability and minimize disruption to regular surgical workflows by affording surgeons the ability to apply stimulation after the procedure, in the recovery room.
The first patient enrolled in REGAIN was treated by Dr. Adil Ladak, a former master’s student of Tessa’s from the University of Alberta, creating a full-circle moment in the journey towards commercializing the first therapeutic solution to improve peripheral nerve injury recovery. "The first patient enrollment is an exciting milestone for Epineuron and witnessing the translation of scientific advancement from bench to bedside is truly remarkable. Our study team is eager to see the progress of the trial as it unfolds,” Chan said.
“We are thrilled to see the REGAIN study come to fruition with the dedication of our entire team. We are committed to forging the path for a better standard of care-for the treatment of injured peripheral nerves,” Epineuron Co-Founder/CEO Sergio Aguirre stated.
Peripulse delivers electrical impulses directly to the damaged nerve during or shortly after surgery. Therapy is administered for an hour, after which the device is removed, and the process of nerve regeneration begins. The treatment is tailored to meet the unique needs of each injury and the preferences of each patient, ensuring personalized care that maximizes comfort and promotes optimal outcomes.
Epineuron is a Canadian clinical-stage nerve care company developing bioelectronic therapy that offers a transformative approach in treating injured peripheral nerves.
The study aims to enroll up to 110 patients across Canada and the United States, making it the largest medical device study for nerve regeneration.
PeriPulse is a direct clinical translation of the research led by Drs. Tessa Gordon and Ming Chan from the University of Alberta spanning basic science to early clinical studies investigating the effects of bioelectronic therapy combined with standard nerve interventions. The proprietary system has been engineered to enhance usability and minimize disruption to regular surgical workflows by affording surgeons the ability to apply stimulation after the procedure, in the recovery room.
The first patient enrolled in REGAIN was treated by Dr. Adil Ladak, a former master’s student of Tessa’s from the University of Alberta, creating a full-circle moment in the journey towards commercializing the first therapeutic solution to improve peripheral nerve injury recovery. "The first patient enrollment is an exciting milestone for Epineuron and witnessing the translation of scientific advancement from bench to bedside is truly remarkable. Our study team is eager to see the progress of the trial as it unfolds,” Chan said.
“We are thrilled to see the REGAIN study come to fruition with the dedication of our entire team. We are committed to forging the path for a better standard of care-for the treatment of injured peripheral nerves,” Epineuron Co-Founder/CEO Sergio Aguirre stated.
Peripulse delivers electrical impulses directly to the damaged nerve during or shortly after surgery. Therapy is administered for an hour, after which the device is removed, and the process of nerve regeneration begins. The treatment is tailored to meet the unique needs of each injury and the preferences of each patient, ensuring personalized care that maximizes comfort and promotes optimal outcomes.
Epineuron is a Canadian clinical-stage nerve care company developing bioelectronic therapy that offers a transformative approach in treating injured peripheral nerves.