Sam Brusco, Associate Editor10.24.23
VUNO, a South Korean medical artificial intelligence (AI) company, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its VUNO Med-DeepBrain AI-powered brain quantification device.
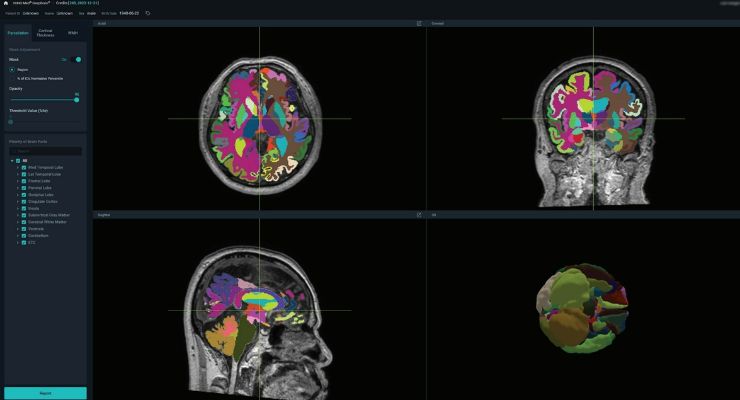
VUNO Med-DeepBrain automates the manual process of identifying, labeling, and quantifying segmentable brain structures from MRI images. The AI software provides volumetric data on more than 100 brain regions through brain parcellation and offers cortical thickness, as well as white matter hyperintensity (WMH).
Atrophy data is compared with a normal population and shown with a normative percentile measurement. The data can be compiled and presented in a customizable report to clinicians, which may be valuable in dementia and other neurodegenerative diseases.
The company plans to enhance collaborations with global pharmaceutical firms that seek AI-based brain MRI quantification technology.
According to clinical research presented at this year’s Alzheimer's Association International Conference (AAIC), VUNO Med-DeepBrain demonstrated its ability to provide information that can be used to predict amyloid positivity in patients experiencing Subjective Cognitive Decline (SCD), one of the earliest noticeable symptoms of Alzheimer's disease and related dementias.
This suggests the product can support early detection and management of patients who may progress to dementia—before mild cognitive impairment (MCI) or early dementia.
VUNO’s CEO Yeha Lee told the press, "VUNO Med-DeepBrain marks the first FDA clearance from VUNO, and we expect it will be a steppingstone for VUNO's expansion into the U.S. market," adding "with this product, we will make every effort to help improve the declining quality of life experienced by many dementia patients."
VUNO Med-DeepBrain automates the manual process of identifying, labeling, and quantifying segmentable brain structures from MRI images. The AI software provides volumetric data on more than 100 brain regions through brain parcellation and offers cortical thickness, as well as white matter hyperintensity (WMH).
Atrophy data is compared with a normal population and shown with a normative percentile measurement. The data can be compiled and presented in a customizable report to clinicians, which may be valuable in dementia and other neurodegenerative diseases.
The company plans to enhance collaborations with global pharmaceutical firms that seek AI-based brain MRI quantification technology.
According to clinical research presented at this year’s Alzheimer's Association International Conference (AAIC), VUNO Med-DeepBrain demonstrated its ability to provide information that can be used to predict amyloid positivity in patients experiencing Subjective Cognitive Decline (SCD), one of the earliest noticeable symptoms of Alzheimer's disease and related dementias.
This suggests the product can support early detection and management of patients who may progress to dementia—before mild cognitive impairment (MCI) or early dementia.
VUNO’s CEO Yeha Lee told the press, "VUNO Med-DeepBrain marks the first FDA clearance from VUNO, and we expect it will be a steppingstone for VUNO's expansion into the U.S. market," adding "with this product, we will make every effort to help improve the declining quality of life experienced by many dementia patients."