Sam Brusco, Associate Editor10.19.23
Edwards Lifesciences has received CE mark approval for its EVOQUE tricuspid valve replacement system. The company says it’s the first transcatheter valve replacement therapy to earn regulatory approval to treat tricuspid regurgitation (TR).
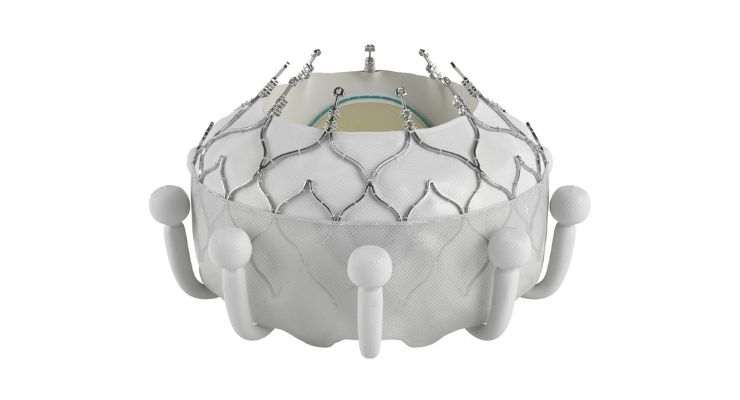
EVOQUE is comprised of a nitinol self-expanding frame, intra-annular sealing skirt, and tissue leaflets made from the company’s bovine pericardial tissue. The valve will be available in three sizes, all delivered though a transfemoral 28F system.
One-year results from Edwards’ TRISCEND study of EVOQUE demonstrated 90.1% survival, 88.4% freedom from heart failure hospitalization, and 97.6% sustained TR reduction to mild or trace TR. There were also functional and quality-of-life improvements—93% of patients in NYHA Class I or II compared to 26% at baseline and a 26-point increase in KCCQ score over baseline.
"Innovating for unmet patient needs is at the center of everything we do at Edwards, which makes us especially proud to have received CE mark for this first-of-its-kind transcatheter tricuspid valve replacement therapy," Daveen Chopra, Edwards' corporate VP, transcatheter mitral and tricuspid therapies told the press. "With the EVOQUE system's approval, in addition to our current PASCAL tricuspid system, we are now able to provide a broader array of much-needed treatment options for appropriate tricuspid disease patients in Europe."
EVOQUE is comprised of a nitinol self-expanding frame, intra-annular sealing skirt, and tissue leaflets made from the company’s bovine pericardial tissue. The valve will be available in three sizes, all delivered though a transfemoral 28F system.
One-year results from Edwards’ TRISCEND study of EVOQUE demonstrated 90.1% survival, 88.4% freedom from heart failure hospitalization, and 97.6% sustained TR reduction to mild or trace TR. There were also functional and quality-of-life improvements—93% of patients in NYHA Class I or II compared to 26% at baseline and a 26-point increase in KCCQ score over baseline.
"Innovating for unmet patient needs is at the center of everything we do at Edwards, which makes us especially proud to have received CE mark for this first-of-its-kind transcatheter tricuspid valve replacement therapy," Daveen Chopra, Edwards' corporate VP, transcatheter mitral and tricuspid therapies told the press. "With the EVOQUE system's approval, in addition to our current PASCAL tricuspid system, we are now able to provide a broader array of much-needed treatment options for appropriate tricuspid disease patients in Europe."