Sam Brusco, Associate Editor09.12.23
CorVista Health, a digital health company aiming to cardiovascular disease diagnosis, has grabbed U.S. Food and Drug Administration (FDA) clearance for its CorVista system with CAD Add-On as a diagnostic aid for significant coronary artery disease.
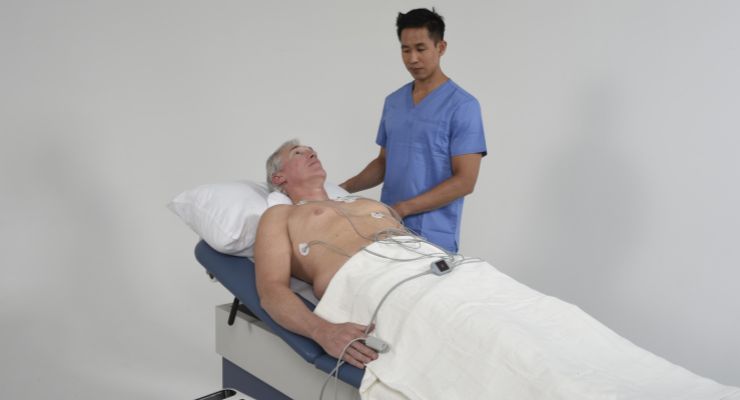
The CorVista system is a non-invasive system of hardware and software components that work together to help physicians evaluate at the point of care for cardiac disease using a static, machine-learned detection algorithm. It’s indicated to analyze sensor-acquired physiological signals of patients that present with cardiovascular symptoms to indicate likelihood of significant coronary artery disease.
The generated analysis is then presented for health provider interpretation along with their clinical judgement, patient signs, symptoms, and clinical history as a diagnostic aid. CorVista said the newly cleared CAD Add-On is the first of its anticipated suite of cardiac detection algorithms to earn market clearance.
“From all of us at CorVista Health, we are thrilled to announce we have obtained FDA clearance of the CorVista System,” Don Crawford, president and CEO of CorVista told the press. “Our point-of-care, non-invasive solution rules out significant CAD with a negative predictive value (NPV) of 99%. Furthermore, it requires no fasting, radiation, or significant capital investment. The ability to obtain a result within minutes will be a game changer in aiding physicians to detect the potential presence of cardiac disease.”
This milestone follows last year’s FDA Breakthrough Designation for the company’s second disease specific Add-On module in development, which is focused on pulmonary hypertension.
“We believe CorVista Health has the ability to transform the way cardiovascular disease is diagnosed, particularly in underserved low-resource settings where there is the greatest need,” said Charles Bridges, MD, ScD., CorVista’s executive VP and chief scientific officer. “CorVista Health’s innovative solution has the potential to make a tremendous impact to our healthcare system and enable more equitable care.”
The CorVista system is a non-invasive system of hardware and software components that work together to help physicians evaluate at the point of care for cardiac disease using a static, machine-learned detection algorithm. It’s indicated to analyze sensor-acquired physiological signals of patients that present with cardiovascular symptoms to indicate likelihood of significant coronary artery disease.
The generated analysis is then presented for health provider interpretation along with their clinical judgement, patient signs, symptoms, and clinical history as a diagnostic aid. CorVista said the newly cleared CAD Add-On is the first of its anticipated suite of cardiac detection algorithms to earn market clearance.
“From all of us at CorVista Health, we are thrilled to announce we have obtained FDA clearance of the CorVista System,” Don Crawford, president and CEO of CorVista told the press. “Our point-of-care, non-invasive solution rules out significant CAD with a negative predictive value (NPV) of 99%. Furthermore, it requires no fasting, radiation, or significant capital investment. The ability to obtain a result within minutes will be a game changer in aiding physicians to detect the potential presence of cardiac disease.”
This milestone follows last year’s FDA Breakthrough Designation for the company’s second disease specific Add-On module in development, which is focused on pulmonary hypertension.
“We believe CorVista Health has the ability to transform the way cardiovascular disease is diagnosed, particularly in underserved low-resource settings where there is the greatest need,” said Charles Bridges, MD, ScD., CorVista’s executive VP and chief scientific officer. “CorVista Health’s innovative solution has the potential to make a tremendous impact to our healthcare system and enable more equitable care.”