Sam Brusco, Associate Editor09.05.23
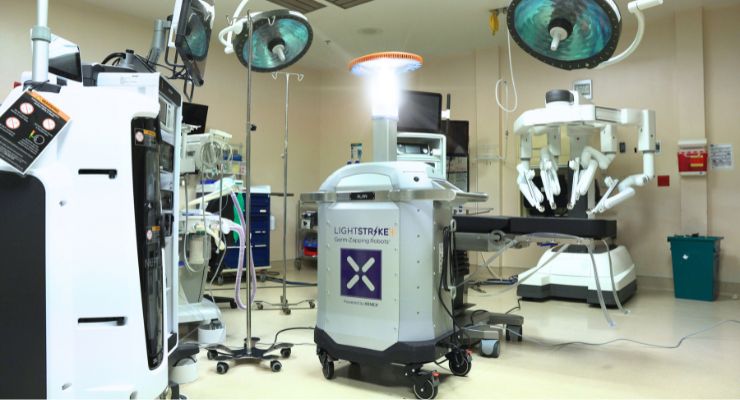
Xenex Disinfection Services has been granted de novo authorization from the U.S. Food and Drug Administration for LightStrike+, the company’s high-intensity, broad-spectrum ultraviolet (UV) light robot.
Xenex said the authorization creates a new medical device product classification, and LightStrike+ is the first, only product of its type. This sets a precedent for FDA’s regulating of UV robots meant to reduce pathogens on non-porous, high-touch surfaces in healthcare.
LightStrike+ aims to perform microbial reduction on non-critical medical device surfaces, free from visual soiling after manual cleaning and disinfection. The UV robot was cleared for healthcare facilities, which covers unoccupied operating rooms, hospital room, and other clinical settings containing non-critical medical devices.
The company said over 1,200 healthcare facilities globally have run LightStrike+ robots over 37 million cycles and its run-time can be as low as two minutes. The UV robot touts 45 peer-reviewed studies showing safe and effective use and 193 patents as well.
LightStrike+ uses a xenon lamp to create high-intensity pulsed UV light that reduces the number of pathogens on surfaces and helps disrupt transmission from one patient or healthcare worker to the next. Xenex said its FDA authorization is supported by rigorous testing performed on over 10,000 samples of vegetative bacteria and Clostridiodes difficile (C.diff) spores.
“We have been working with our hospital partners for over a decade to support them in their mission to improve patient safety and public health. As an infectious diseases epidemiologist, I am highly concerned about antibiotic resistance in the hospital environment. I hope that FDA authorization will allow hospitals to more easily use this tool in their fight against pathogens,” Dr. Mark “Tuck” Stibich, founder and chief scientific officer of Xenex told the press.
“We understand that it can be challenging for hospitals to evaluate UV technologies, especially given the unverified and often exaggerated claims made by some manufacturers. FDA authorization gives hospital decision-makers the confidence that Xenex’s claims for LightStrike+ are accurate and validated,” added Xenex CEO Morris Miller.
Xenex said the authorization creates a new medical device product classification, and LightStrike+ is the first, only product of its type. This sets a precedent for FDA’s regulating of UV robots meant to reduce pathogens on non-porous, high-touch surfaces in healthcare.
LightStrike+ aims to perform microbial reduction on non-critical medical device surfaces, free from visual soiling after manual cleaning and disinfection. The UV robot was cleared for healthcare facilities, which covers unoccupied operating rooms, hospital room, and other clinical settings containing non-critical medical devices.
The company said over 1,200 healthcare facilities globally have run LightStrike+ robots over 37 million cycles and its run-time can be as low as two minutes. The UV robot touts 45 peer-reviewed studies showing safe and effective use and 193 patents as well.
LightStrike+ uses a xenon lamp to create high-intensity pulsed UV light that reduces the number of pathogens on surfaces and helps disrupt transmission from one patient or healthcare worker to the next. Xenex said its FDA authorization is supported by rigorous testing performed on over 10,000 samples of vegetative bacteria and Clostridiodes difficile (C.diff) spores.
“We have been working with our hospital partners for over a decade to support them in their mission to improve patient safety and public health. As an infectious diseases epidemiologist, I am highly concerned about antibiotic resistance in the hospital environment. I hope that FDA authorization will allow hospitals to more easily use this tool in their fight against pathogens,” Dr. Mark “Tuck” Stibich, founder and chief scientific officer of Xenex told the press.
“We understand that it can be challenging for hospitals to evaluate UV technologies, especially given the unverified and often exaggerated claims made by some manufacturers. FDA authorization gives hospital decision-makers the confidence that Xenex’s claims for LightStrike+ are accurate and validated,” added Xenex CEO Morris Miller.