Sam Brusco, Associate Editor08.10.23
The June 5 recall of Datascope/Maquet/Getinge’s Cardiosave Hybrid intra-balloon pump (IABP) and Cardiosave Rescue IABP) has been identified as Class I by the U.S. Food and Drug Administration (FDA).
The company recalled the IABPs over unexpected shutdown due to electrical failures in their power management boards or solenoid boards. It warned of possible unstable blood pressure, inadequate blood supply, a vital organ injury, or death due to the issue.
According to the FDA, Getinge reported 26 complaints. There are not yet reports of injuries or deaths due to the issue.
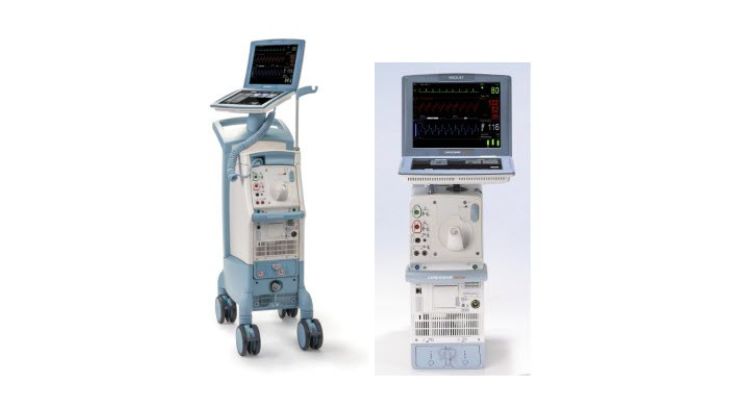
Cardioscave Hybrid and Rescue IABPs are electromechanical systems to inflate and deflate intra-aortic balloons. They offer temporary support to the left ventricle using counter pulsation. Once the balloon is positioned in the aorta, the pump works in synchrony with the ECG or arterial pressure waveform to make the balloon inflate and deflate at the correct time during the cardiac cycle.
Cardiosave IABPs are indicated for acute coronary syndrome, cardiac and non-cardiac surgery, or complications of heart failure in adults.
Getinge advised customers to ensure there’s an alternative IABP available to support therapy, and offer hemodynamic support if there’s no other means to continue counterpulsation therapy.
The company recalled the IABPs over unexpected shutdown due to electrical failures in their power management boards or solenoid boards. It warned of possible unstable blood pressure, inadequate blood supply, a vital organ injury, or death due to the issue.
According to the FDA, Getinge reported 26 complaints. There are not yet reports of injuries or deaths due to the issue.
Cardioscave Hybrid and Rescue IABPs are electromechanical systems to inflate and deflate intra-aortic balloons. They offer temporary support to the left ventricle using counter pulsation. Once the balloon is positioned in the aorta, the pump works in synchrony with the ECG or arterial pressure waveform to make the balloon inflate and deflate at the correct time during the cardiac cycle.
Cardiosave IABPs are indicated for acute coronary syndrome, cardiac and non-cardiac surgery, or complications of heart failure in adults.
Getinge advised customers to ensure there’s an alternative IABP available to support therapy, and offer hemodynamic support if there’s no other means to continue counterpulsation therapy.