Sam Brusco, Associate Editor07.31.23
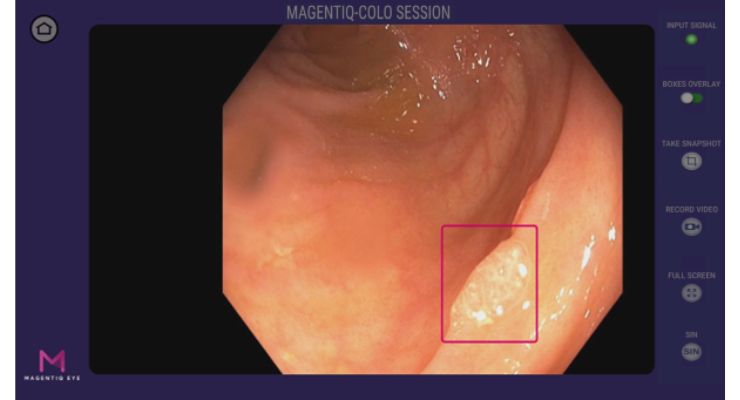
MAGENTIQ-EYE has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its MAGENTIQ-COLO, an artificial intelligence (AI) solution for colonoscopy.
The company said the device offers an increase in adenoma detection rate (ADR) for the gastroenterology community. A 2022 study in 10 leading medical centers with 29 endoscopy experts and over 950 patients validated MAGENTIQ-COLO as one of the best performing AI solutions in the category.
According to MAGENTIQ-EYE, ADR was increased by 26% relatively (7% in absolute values), which translates into a 21% decrease in colorectal cancer (CRC) and 35% decrease in patient mortality.
"FDA clearance is a major milestone, and we are very proud to join only a handful of companies in the field of AI-aided colonoscopy to be granted clearance. Our next step will be to pursue opportunities in the U.S. market as we look forward to increasing the accuracy of detecting early signs of CRC and taking part in saving the lives of thousands of Americans," Dror Zur, founder and CEO of MAGENTIQ-EYE, told the press. "Although the sense of accomplishment is strong, I believe this is only the tip of the iceberg for AI in gastroenterology, so stay tuned for new products and features from us coming soon."
The company said the device offers an increase in adenoma detection rate (ADR) for the gastroenterology community. A 2022 study in 10 leading medical centers with 29 endoscopy experts and over 950 patients validated MAGENTIQ-COLO as one of the best performing AI solutions in the category.
According to MAGENTIQ-EYE, ADR was increased by 26% relatively (7% in absolute values), which translates into a 21% decrease in colorectal cancer (CRC) and 35% decrease in patient mortality.
"FDA clearance is a major milestone, and we are very proud to join only a handful of companies in the field of AI-aided colonoscopy to be granted clearance. Our next step will be to pursue opportunities in the U.S. market as we look forward to increasing the accuracy of detecting early signs of CRC and taking part in saving the lives of thousands of Americans," Dror Zur, founder and CEO of MAGENTIQ-EYE, told the press. "Although the sense of accomplishment is strong, I believe this is only the tip of the iceberg for AI in gastroenterology, so stay tuned for new products and features from us coming soon."