Sam Brusco, Associate Editor07.10.23
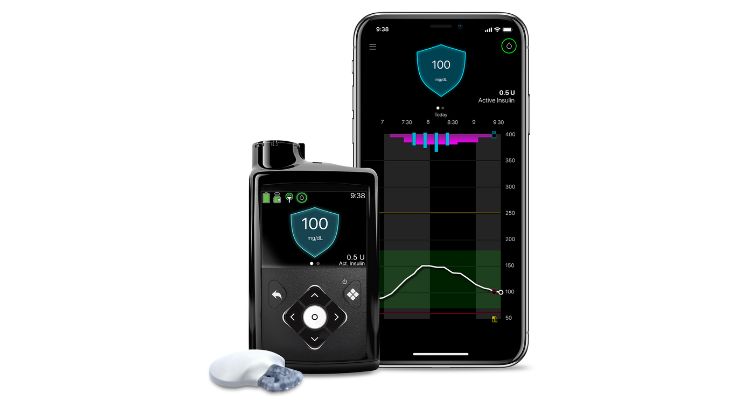
Medtronic announced that its MiniMed 780G automated insulin pump with new Guardian 4 continuous glucose monitoring (CGM) sensor is now covered for all eligible Medicare and Medicare Advantage beneficiaries.
The Guardian 4 sensor, according to Medtronic, doesn’t need fingersticks while in SmartGuard technology. The company has begun processing orders and will begin shipping in the next few weeks to type 1 diabetes patients meeting eligibility criteria.
“Coming off the heels of a very exciting 83rd American Diabetes Association Scientific Sessions where we shared the latest real-world data on our MiniMed 780G system, we couldn’t be more thrilled to expand access to our Meal Detection technology that’s helping many individuals around the world achieve an average Time in Range exceeding 80%1 when using recommended settings,” Que Dallara, EVP and president of Medtronic Diabetes told the press. “We see people of all ages and from different therapies benefitting significantly from improved outcomes with the MiniMed 780G system. We attribute this to the easy-to-use design coupled with a powerful algorithm that provides coverage when life gets in the way—one that’s designed for real life.”
MiniMed 780G provides automatic blood sugar adjustments and corrections every five minutes to help those who occasionally forget to bolus or underestimate the number of carbs in their meal. It features a low target setting—as low as 100 mg/dL to more closely match the average glucose of someone without diabetes.
“What’s particularly compelling about the MiniMed 780G system is that it broadens eligibility to many more individuals interested in automated insulin delivery to help them achieve their diabetes management goals,” said Robert Vigersky, MD, chief medical officer of Medtronic Diabetes. “Historically, there’s been a stepwise approach towards prescribing pump therapy when certain criteria were met including the ability to carb count and use advanced technology. This approach is no longer relevant given the system’s ability to overcome these challenges. We are very pleased that we’re able to make this technology available to Medicare beneficiaries.”
MiniMed 780G is approved in the U.S. for those with type 1 diabetes aged seven and above.
Reference
1 Medtronic data on file: MiniMed 780G data uploaded voluntarily by 135 users in US, as of June 19, 2023. Results reflect users using recommended settings. Real-World clinical data presented here refer to MiniMed 780G system used in combination with Guardian sensor 3 and/or Guardian 4 sensor.
The Guardian 4 sensor, according to Medtronic, doesn’t need fingersticks while in SmartGuard technology. The company has begun processing orders and will begin shipping in the next few weeks to type 1 diabetes patients meeting eligibility criteria.
“Coming off the heels of a very exciting 83rd American Diabetes Association Scientific Sessions where we shared the latest real-world data on our MiniMed 780G system, we couldn’t be more thrilled to expand access to our Meal Detection technology that’s helping many individuals around the world achieve an average Time in Range exceeding 80%1 when using recommended settings,” Que Dallara, EVP and president of Medtronic Diabetes told the press. “We see people of all ages and from different therapies benefitting significantly from improved outcomes with the MiniMed 780G system. We attribute this to the easy-to-use design coupled with a powerful algorithm that provides coverage when life gets in the way—one that’s designed for real life.”
MiniMed 780G provides automatic blood sugar adjustments and corrections every five minutes to help those who occasionally forget to bolus or underestimate the number of carbs in their meal. It features a low target setting—as low as 100 mg/dL to more closely match the average glucose of someone without diabetes.
“What’s particularly compelling about the MiniMed 780G system is that it broadens eligibility to many more individuals interested in automated insulin delivery to help them achieve their diabetes management goals,” said Robert Vigersky, MD, chief medical officer of Medtronic Diabetes. “Historically, there’s been a stepwise approach towards prescribing pump therapy when certain criteria were met including the ability to carb count and use advanced technology. This approach is no longer relevant given the system’s ability to overcome these challenges. We are very pleased that we’re able to make this technology available to Medicare beneficiaries.”
MiniMed 780G is approved in the U.S. for those with type 1 diabetes aged seven and above.
Reference
1 Medtronic data on file: MiniMed 780G data uploaded voluntarily by 135 users in US, as of June 19, 2023. Results reflect users using recommended settings. Real-World clinical data presented here refer to MiniMed 780G system used in combination with Guardian sensor 3 and/or Guardian 4 sensor.