Michael Barbella, Managing Editor07.05.23
The structural heart occlusion (SHO) devices market has experienced significant growth in recent years—25% annually from 2015-2022. Driven mainly by Boston Scientific’s Watchman FLX device, this sector expanded more than 150% last year compared with 2019, far outpacing other cardiovascular device markets. The SHO devices market is forecast to exceed $7 billion by 2033, according to GlobalData.
“Physicians implant the Watchman FLX device in the left atrial appendage (LAA) of patients with atrial fibrillation to prevent the formation of a blood clot that would lead to a stroke," GlobalData Medical Devices Analyst Joselia Carlos said. "Physicians typically prescribe blood-thinners first before resorting to LAA implanted occlusion devices (e.g., the Watchman FLX). However, if patients have contraindications against blood-thinners, LAA implanted occlusion devices then become the recommended treatment option.”
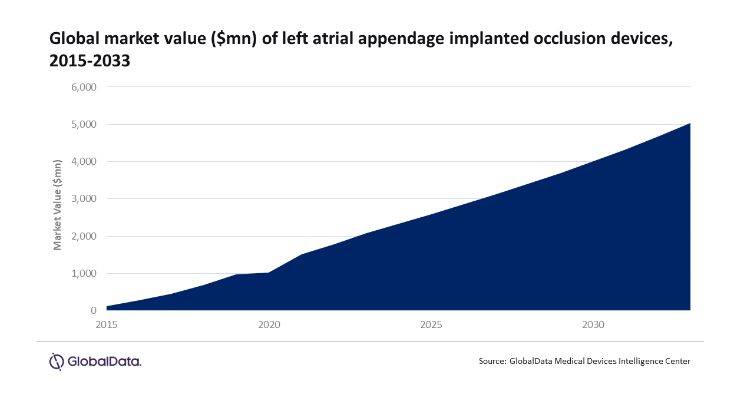
The market value for LAA implanted occlusion devices mushroomed from $132 million in 2015 to $1.79 billion in 2022. According to Boston Scientific’s 2022 annual report, the Watchman FLX has been implanted in more than 300,000 patients since its U.S. Food and Drug Administration and CE mark approvals, both in 2019. As a result of the Watchman FLX, Boston Scientific currently holds a 94% market share of the LAA implanted occlusion device market.
“At one point, the version prior to the Watchman FLX, the Watchman 2.5, practically had 100% market share because it was the only LAA implanted occlusion device that was FDA-approved at the time. Then, in 2021, Abbott gained FDA approval for their LAA implanted occlusion device—the Amplatzer Amulet," Carlos noted. "While Abbott successfully gained some market share, Boston Scientific’s Watchman device remains the number one device for LAA implanted closure procedures.”
While the Watchman device is widely used in the United States and Europe, this exponential growth cannot be assumed in other countries once the Watchman becomes commercially available in those nations. For example, since the Watchman device’s introduction in India in 2017, only about 50 LAA closure procedures have been completed, according to GlobalData’s estimates.
“There is a strong opposition against the use of the Watchman device amongst doctors in India because of three reasons—the procedure is expensive, associated adverse events, and the procedure’s complexity. Even though the Watchman device has been incredibly successful in the U.S. and Europe, it has yet to break through in some countries,” Carlos stated.
“Physicians implant the Watchman FLX device in the left atrial appendage (LAA) of patients with atrial fibrillation to prevent the formation of a blood clot that would lead to a stroke," GlobalData Medical Devices Analyst Joselia Carlos said. "Physicians typically prescribe blood-thinners first before resorting to LAA implanted occlusion devices (e.g., the Watchman FLX). However, if patients have contraindications against blood-thinners, LAA implanted occlusion devices then become the recommended treatment option.”
The market value for LAA implanted occlusion devices mushroomed from $132 million in 2015 to $1.79 billion in 2022. According to Boston Scientific’s 2022 annual report, the Watchman FLX has been implanted in more than 300,000 patients since its U.S. Food and Drug Administration and CE mark approvals, both in 2019. As a result of the Watchman FLX, Boston Scientific currently holds a 94% market share of the LAA implanted occlusion device market.
“At one point, the version prior to the Watchman FLX, the Watchman 2.5, practically had 100% market share because it was the only LAA implanted occlusion device that was FDA-approved at the time. Then, in 2021, Abbott gained FDA approval for their LAA implanted occlusion device—the Amplatzer Amulet," Carlos noted. "While Abbott successfully gained some market share, Boston Scientific’s Watchman device remains the number one device for LAA implanted closure procedures.”
While the Watchman device is widely used in the United States and Europe, this exponential growth cannot be assumed in other countries once the Watchman becomes commercially available in those nations. For example, since the Watchman device’s introduction in India in 2017, only about 50 LAA closure procedures have been completed, according to GlobalData’s estimates.
“There is a strong opposition against the use of the Watchman device amongst doctors in India because of three reasons—the procedure is expensive, associated adverse events, and the procedure’s complexity. Even though the Watchman device has been incredibly successful in the U.S. and Europe, it has yet to break through in some countries,” Carlos stated.