Sam Brusco, Associate Editor06.28.23
INVO Bioscience, a fertility company, has received U.S. Food and Drug Administration (FDA) 510(k) clearance to expand labeling on its INVOcell device and indication for use to provide for a five-day incubation period.
The INVOcell device allows fertilization and early embryo development to take place in vivo within the woman’s body. It’s the first intravaginal culture (IVC) technique to incubate oocytes and sperm during fertilization and early embryo development.
"This is a momentous day for INVO as the FDA has provided clearance for us to expand our labeling to cover a 5-day incubation period for INVOcell," Steve Shum, CEO of INVO told the press. "This has been a multi-year effort to demonstrate INVOcell's ability to improve patient outcomes using a longer incubation period, similar to conventional IVF results. We believe our ability to now communicate the improved success rates using INVOcell to patients and physicians will have a positive effect on the overall confidence and adoption of the technology going forward."
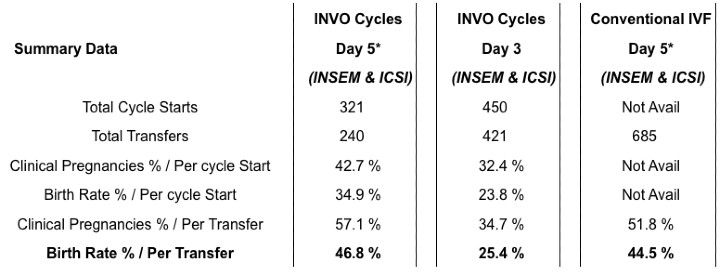
Retrospective (real-world data) collected from four separate clinics (2017-2019).
The IVC technique offers a more natural, intimate, and affordable experience compared to other treatments, and the company believes it can deliver comparable results at a fraction of the price of traditional in vitro fertilization (IVF).
"INVOcell originally received De Novo clearance from the FDA for a day 3 incubation period," continued Shum. "Over time, industry trends for conventional IVF have moved more towards a day 5 incubation period which has generally improved pregnancy success rates. INVOcell was often being used off-label for 5-day, which similarly showed improved outcomes, and which afforded the opportunity to use real market usage data to support our 510k submission. Today's 510(k) clearance by the FDA for INVOcell use with 5-day incubation is a major accomplishment and now allows us to better showcase what INVOcell and the IVC procedure can deliver to patients in need of an affordable fertility solution. We sincerely appreciate the significant effort by our team and the clinics that contributed their real-world data."
INVO’s approach includes opening dedicated "INVO Centers" offering INVOcell and IVC procedure (three centers in North America now operational), acquisition of existing profitable IVF clinics (signed binding agreements to acquire Wisconsin Fertility Institute), and continued global distribution and sale of INVOcell into existing fertility clinics.
The INVOcell device allows fertilization and early embryo development to take place in vivo within the woman’s body. It’s the first intravaginal culture (IVC) technique to incubate oocytes and sperm during fertilization and early embryo development.
"This is a momentous day for INVO as the FDA has provided clearance for us to expand our labeling to cover a 5-day incubation period for INVOcell," Steve Shum, CEO of INVO told the press. "This has been a multi-year effort to demonstrate INVOcell's ability to improve patient outcomes using a longer incubation period, similar to conventional IVF results. We believe our ability to now communicate the improved success rates using INVOcell to patients and physicians will have a positive effect on the overall confidence and adoption of the technology going forward."

Retrospective (real-world data) collected from four separate clinics (2017-2019).
The IVC technique offers a more natural, intimate, and affordable experience compared to other treatments, and the company believes it can deliver comparable results at a fraction of the price of traditional in vitro fertilization (IVF).
"INVOcell originally received De Novo clearance from the FDA for a day 3 incubation period," continued Shum. "Over time, industry trends for conventional IVF have moved more towards a day 5 incubation period which has generally improved pregnancy success rates. INVOcell was often being used off-label for 5-day, which similarly showed improved outcomes, and which afforded the opportunity to use real market usage data to support our 510k submission. Today's 510(k) clearance by the FDA for INVOcell use with 5-day incubation is a major accomplishment and now allows us to better showcase what INVOcell and the IVC procedure can deliver to patients in need of an affordable fertility solution. We sincerely appreciate the significant effort by our team and the clinics that contributed their real-world data."
INVO’s approach includes opening dedicated "INVO Centers" offering INVOcell and IVC procedure (three centers in North America now operational), acquisition of existing profitable IVF clinics (signed binding agreements to acquire Wisconsin Fertility Institute), and continued global distribution and sale of INVOcell into existing fertility clinics.












