Michael Barbella, Managing Editor06.27.23
The Flume Catheter Company Ltd. has launched a study to gather real-world data on patient experiences with the new FLUME catheter.
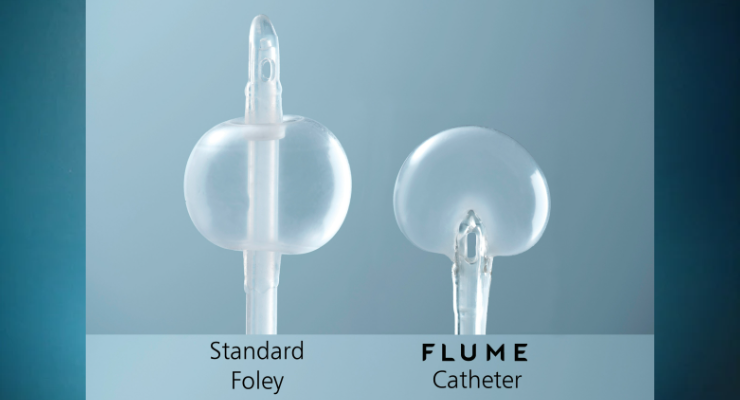
Cleared in November 2021 by the U.S. Food and Drug Administration (FDA), the catheter is designed to address some of the triggers that can negatively impact patients’ comfort and quality of life. The FLUME catheter is distinguished by an alternative balloon configuration, designed to envelop the tip of the catheter upon inflation and, thereby, allow the emptying bladder to come into contact with the compliant balloon rather than a rigid catheter tip. The drainage holes are inset low in the balloon, designed to enhance bladder drainage as well as guard against bladder mucosa being drawn into the drainage holes.
Researchers at Duke Clinical Research Institute and Penn State College of Medicine have formed a strategic partnership to conduct the study and obtain first-hand information from patients with indwelling catheters. Participants will provide their feedback before and after using the FLUME catheter for up to 30 days, which will allow researchers to assess patients’ perceptions around comfort and quality of life factors with their traditional catheters and with the new FLUME catheter.
“The FLUME design eliminates the exposed catheter tip, which has been a feature of nearly all urethral catheters since Dr. Foley’s original model in the 1930s,” said Charles Scales, M.D., FLUME Catheter Comfort Study principal investigator, and associate professor of Surgery in the Division of Urology at Duke University School of Medicine. “We know that around 50% of patients report discomfort, and we are excited to partner with FLUME to evaluate patient comfort among people who need catheters to empty their bladders. This could be game-changer for these patients.”
The new study will help researchers learn more about whether the FLUME catheter may improve patients’ perceptions around comfort and quality of life. The study is taking place at the Penn State Health Urology clinic on the Penn State Health Milton S. Hershey Medical Center campus and will last for approximately six months. Patients who are 18 years of age or older and have used an indwelling catheter for at least three months may be able to participate.
“We are delighted to be embarking on this study to measure the impact of the FLUME design on patients’ comfort and quality of life,” The Flume Catheter Company Chairman/Chief Medical Officer Dr. John Havard said. “Our goal—and the inspiration behind the FLUME catheter—is to transform the lives of the 100 million patients worldwide who rely on indwelling urinary catheters. We expect to gain valuable insights from this study.”
The Flume Catheter Company was founded in the United Kongdom in 2016 by Havard and engineer, private equity investor and former M&S CEO Roger Holmes. The firm’s mission is to improve catheter design to enhance patient experience and outcomes and reduce the catheter-associated healthcare cost burden.
Located on the campus of Penn State Health Milton S. Hershey Medical Center in Hershey, Pa., Penn State College of Medicine boasts a portfolio of more than $150 million in funded research annually. Projects range from development of artificial organs and advanced diagnostics to groundbreaking cancer treatments and understanding the fundamental causes of disease. Enrolling its first students in 1967, the College of Medicine has more than 1,700 students and trainees in medicine, nursing, other health professions and biomedical research in both Hershey and State College, Pa. The College of Medicine shares an integrated strategic plan and operations with Penn State Health.
The Duke Clinical Research Institute, part of the Duke University School of Medicine, is the largest academic clinical research organization in the world. It develops, shares, and implements knowledge that improves global health through clinical research. The institute conducts multinational clinical trials, manages major national patient registries, and performs landmark outcomes research. The DCRI is a pioneer in cardiovascular and pediatric clinical research, and conducts clinical research across multiple other therapeutic areas, including urology, musculoskeletal pain and surgical sciences, infectious disease, neuroscience, respiratory medicine, and nephrology.
Cleared in November 2021 by the U.S. Food and Drug Administration (FDA), the catheter is designed to address some of the triggers that can negatively impact patients’ comfort and quality of life. The FLUME catheter is distinguished by an alternative balloon configuration, designed to envelop the tip of the catheter upon inflation and, thereby, allow the emptying bladder to come into contact with the compliant balloon rather than a rigid catheter tip. The drainage holes are inset low in the balloon, designed to enhance bladder drainage as well as guard against bladder mucosa being drawn into the drainage holes.
Researchers at Duke Clinical Research Institute and Penn State College of Medicine have formed a strategic partnership to conduct the study and obtain first-hand information from patients with indwelling catheters. Participants will provide their feedback before and after using the FLUME catheter for up to 30 days, which will allow researchers to assess patients’ perceptions around comfort and quality of life factors with their traditional catheters and with the new FLUME catheter.
“The FLUME design eliminates the exposed catheter tip, which has been a feature of nearly all urethral catheters since Dr. Foley’s original model in the 1930s,” said Charles Scales, M.D., FLUME Catheter Comfort Study principal investigator, and associate professor of Surgery in the Division of Urology at Duke University School of Medicine. “We know that around 50% of patients report discomfort, and we are excited to partner with FLUME to evaluate patient comfort among people who need catheters to empty their bladders. This could be game-changer for these patients.”
The new study will help researchers learn more about whether the FLUME catheter may improve patients’ perceptions around comfort and quality of life. The study is taking place at the Penn State Health Urology clinic on the Penn State Health Milton S. Hershey Medical Center campus and will last for approximately six months. Patients who are 18 years of age or older and have used an indwelling catheter for at least three months may be able to participate.
“We are delighted to be embarking on this study to measure the impact of the FLUME design on patients’ comfort and quality of life,” The Flume Catheter Company Chairman/Chief Medical Officer Dr. John Havard said. “Our goal—and the inspiration behind the FLUME catheter—is to transform the lives of the 100 million patients worldwide who rely on indwelling urinary catheters. We expect to gain valuable insights from this study.”
The Flume Catheter Company was founded in the United Kongdom in 2016 by Havard and engineer, private equity investor and former M&S CEO Roger Holmes. The firm’s mission is to improve catheter design to enhance patient experience and outcomes and reduce the catheter-associated healthcare cost burden.
Located on the campus of Penn State Health Milton S. Hershey Medical Center in Hershey, Pa., Penn State College of Medicine boasts a portfolio of more than $150 million in funded research annually. Projects range from development of artificial organs and advanced diagnostics to groundbreaking cancer treatments and understanding the fundamental causes of disease. Enrolling its first students in 1967, the College of Medicine has more than 1,700 students and trainees in medicine, nursing, other health professions and biomedical research in both Hershey and State College, Pa. The College of Medicine shares an integrated strategic plan and operations with Penn State Health.
The Duke Clinical Research Institute, part of the Duke University School of Medicine, is the largest academic clinical research organization in the world. It develops, shares, and implements knowledge that improves global health through clinical research. The institute conducts multinational clinical trials, manages major national patient registries, and performs landmark outcomes research. The DCRI is a pioneer in cardiovascular and pediatric clinical research, and conducts clinical research across multiple other therapeutic areas, including urology, musculoskeletal pain and surgical sciences, infectious disease, neuroscience, respiratory medicine, and nephrology.