Sam Brusco, Associate Editor04.18.23
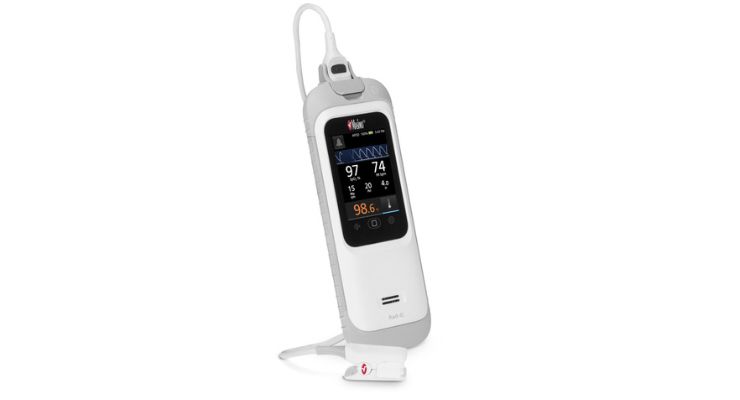
Masimo has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its Rad-G with Temperature, a handheld monitor that offers SET pulse oximetry, respiration rate from the pleth (RRp), and other parameters along with non-contact infrared clinical thermometry.
Rad-G with Temperature aims to make it easier to quickly measure vital signs using a single, compact, portable device. It’s designed for use in physicians’ offices, outpatient services, urgent care facilities, and in first-responder scenarios both indoors and in the field.
With Rad-G, we set out to create an accessible, high-quality care solution that could be used in a multitude of care settings to serve the five billion people on our planet that to date have not had access to pulse oximetry, let alone SET pulse oximetry,” Joe Kiani, founder and CEO of Masimo told the press. “With the addition of temperature measurements, Rad-G is more versatile than ever, streamlining the assessment of multiple key vital signs. Having a product that is light, small, multifunctional, and ‘accurate when you need it most’ is crucial, and Rad-G was designed to be just that. With this FDA clearance, Rad-G with Temperature can now be deployed across the U.S., in addition to many other parts of the world, helping support clinicians in almost any care scenario.”
The thermometer doesn’t need probe covers or other disposable accessories and removes the need to find a separate clinical thermometer to take body temperature measurements. Rad-G’s rechargeable battery offers 24 hours of operation between charges.
Rad-G with Temperature was first developed in collaboration with The Bill & Melinda Gates Foundation as a spot-check device for pneumonia screening. It originally launched outside the U.S. and since European introduction has been adopted by more than 100 global customers. The National Health Service (NHS) England uses the tech to support primary care assessments of unwell children.
The same SpO2 sensor can be used to monitor respiration rate from the plethysmograph, with RRp. Difficulty breathing and fever are considered two of the earliest signs of patient deterioration. Rad-G with Temperature can be used with a variety of reusable and single-patient use sensors. The universal direct-connect Rad-G reusable sensor, indicated for monitoring adult, pediatric, and infant patients, helps eliminate the need to stock and carry multiple sensor types, increasing versatility and ease of use, especially in more challenging field environments.
Earlier this month, Masimo was granted de novo authorization from the FDA for its Opioid Halo opioid overdose prevention and alert system.
Rad-G with Temperature aims to make it easier to quickly measure vital signs using a single, compact, portable device. It’s designed for use in physicians’ offices, outpatient services, urgent care facilities, and in first-responder scenarios both indoors and in the field.
With Rad-G, we set out to create an accessible, high-quality care solution that could be used in a multitude of care settings to serve the five billion people on our planet that to date have not had access to pulse oximetry, let alone SET pulse oximetry,” Joe Kiani, founder and CEO of Masimo told the press. “With the addition of temperature measurements, Rad-G is more versatile than ever, streamlining the assessment of multiple key vital signs. Having a product that is light, small, multifunctional, and ‘accurate when you need it most’ is crucial, and Rad-G was designed to be just that. With this FDA clearance, Rad-G with Temperature can now be deployed across the U.S., in addition to many other parts of the world, helping support clinicians in almost any care scenario.”
The thermometer doesn’t need probe covers or other disposable accessories and removes the need to find a separate clinical thermometer to take body temperature measurements. Rad-G’s rechargeable battery offers 24 hours of operation between charges.
Rad-G with Temperature was first developed in collaboration with The Bill & Melinda Gates Foundation as a spot-check device for pneumonia screening. It originally launched outside the U.S. and since European introduction has been adopted by more than 100 global customers. The National Health Service (NHS) England uses the tech to support primary care assessments of unwell children.
The same SpO2 sensor can be used to monitor respiration rate from the plethysmograph, with RRp. Difficulty breathing and fever are considered two of the earliest signs of patient deterioration. Rad-G with Temperature can be used with a variety of reusable and single-patient use sensors. The universal direct-connect Rad-G reusable sensor, indicated for monitoring adult, pediatric, and infant patients, helps eliminate the need to stock and carry multiple sensor types, increasing versatility and ease of use, especially in more challenging field environments.
Earlier this month, Masimo was granted de novo authorization from the FDA for its Opioid Halo opioid overdose prevention and alert system.