Sam Brusco, Associate Editor03.21.23
SurgVision, a company developing solutions for fluorescence-guided surgical and interventional oncology, has achieved U.S. Food and Drug Administration (FDA) 510(k) clearance for its EXPLORER AIR II for use with Pafolacianine (CYTALUX, On Target Laboratories Inc.) during intraoperative fluorescence imaging.
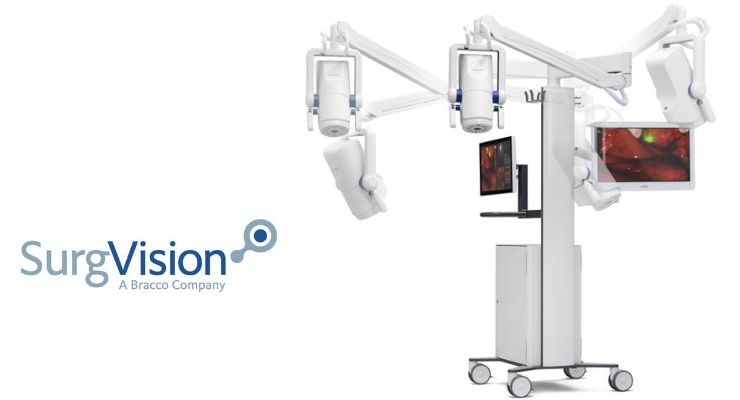
The first product based on the company’s technology platform, it’s designed to achieve high sensitivity and imaging fidelity for oncological intra-op fluorescence imaging. The system provides real-time imaging during surgery and its prototype was testing by academic centers for a number of indications.
"We are very excited about this important milestone. Our goal is to make the EXPLORER AIR II available to surgeons supporting them in their mission to fight cancer," Stefan Schorling, CEO of SurgVision told the press.
EXPLORER AIR II is cleared in the U.S. and EU for visually assessing blood flow and tissue perfusion.
Currently, identifying tumors during surgery of interventional endoscopy relies on visual inspection and palpation. However, since tumor tissue is sometimes difficult to distinguish from healthy tissue, surgical tumor resection is often incomplete.
SurgVision responded with a development pipeline combining highly sensitive imaging to visualize tumors during surgical or interventional procedures in real-time.
The first product based on the company’s technology platform, it’s designed to achieve high sensitivity and imaging fidelity for oncological intra-op fluorescence imaging. The system provides real-time imaging during surgery and its prototype was testing by academic centers for a number of indications.
"We are very excited about this important milestone. Our goal is to make the EXPLORER AIR II available to surgeons supporting them in their mission to fight cancer," Stefan Schorling, CEO of SurgVision told the press.
EXPLORER AIR II is cleared in the U.S. and EU for visually assessing blood flow and tissue perfusion.
Currently, identifying tumors during surgery of interventional endoscopy relies on visual inspection and palpation. However, since tumor tissue is sometimes difficult to distinguish from healthy tissue, surgical tumor resection is often incomplete.
SurgVision responded with a development pipeline combining highly sensitive imaging to visualize tumors during surgical or interventional procedures in real-time.