Sam Brusco, Associate Editor03.17.23
Prolira, a company specializing in treating patients at risk of acute brain failure, has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for its DeltaScan brain state monitor to help diagnose acute encephalopathy in hospitalized patients over 60.
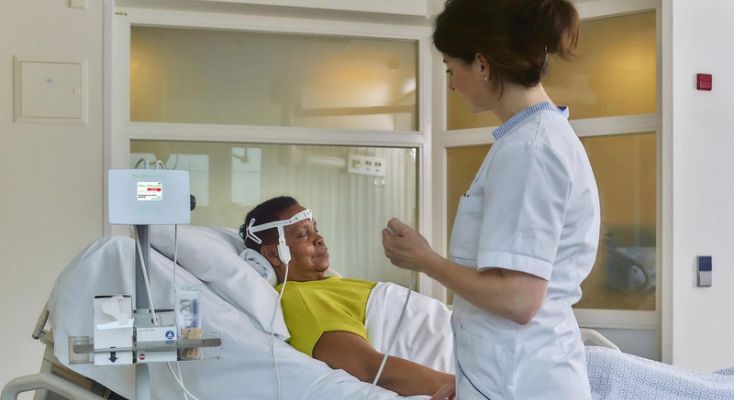
DeltaScan offers quick and objective measurement of acute brain failure even before symptoms appear so immediate treatment can occur and patients can retain their cognitive well-being.
“Managing acute encephalopathy is challenging as symptoms are often overlooked and the diagnostic approaches are largely subjective and ineffective, resulting in delayed diagnosis and treatment. This delay often leads to cognitive impairment, worse health outcomes, and increased costs,” Dr. Frans Leijten, Head of EEG Department, UMC Utrecht, the Netherlands, told the press. “DeltaScan enables clinicians to objectively identify acute encephalopathy, which encompasses delirium, alerting us to act quickly to determine the underlying cause and appropriate interventions. It’s clear from the evidence that minimizing the duration of acute brain failure can improve outcomes.”
DeltaScan is the first bedside platform engineered to help objectively determine acute brain failure before symptoms occur by measuring polymorphic delta waves in the patient’s electroencephalogram (EEG). Nurses or technicians can perform a bedside measurement and get a clear negative or positive output in under five minutes without needing specialized personnel or equipment.
According to Prolia, in a study of over 400 ICU and post-op patients (mostly over 60) showed diagnostic performance of more than 85% for acute encephalopathy.
“FDA clearance is a major milestone in our mission to make brain state assessments and followup interventions an integral part of standard practice. The uptake of DeltaScan in Europe has been strong and has allowed the company to incorporate clinician feedback into the product design, generate robust clinical evidence, and refine our commercial approach,” said Dominic Spadafore, CEO of Prolira. “We’re excited to bring our unique solution to the U.S.”
DeltaScan offers quick and objective measurement of acute brain failure even before symptoms appear so immediate treatment can occur and patients can retain their cognitive well-being.
“Managing acute encephalopathy is challenging as symptoms are often overlooked and the diagnostic approaches are largely subjective and ineffective, resulting in delayed diagnosis and treatment. This delay often leads to cognitive impairment, worse health outcomes, and increased costs,” Dr. Frans Leijten, Head of EEG Department, UMC Utrecht, the Netherlands, told the press. “DeltaScan enables clinicians to objectively identify acute encephalopathy, which encompasses delirium, alerting us to act quickly to determine the underlying cause and appropriate interventions. It’s clear from the evidence that minimizing the duration of acute brain failure can improve outcomes.”
DeltaScan is the first bedside platform engineered to help objectively determine acute brain failure before symptoms occur by measuring polymorphic delta waves in the patient’s electroencephalogram (EEG). Nurses or technicians can perform a bedside measurement and get a clear negative or positive output in under five minutes without needing specialized personnel or equipment.
According to Prolia, in a study of over 400 ICU and post-op patients (mostly over 60) showed diagnostic performance of more than 85% for acute encephalopathy.
“FDA clearance is a major milestone in our mission to make brain state assessments and followup interventions an integral part of standard practice. The uptake of DeltaScan in Europe has been strong and has allowed the company to incorporate clinician feedback into the product design, generate robust clinical evidence, and refine our commercial approach,” said Dominic Spadafore, CEO of Prolira. “We’re excited to bring our unique solution to the U.S.”