Michael Barbella, Managing Editor02.15.23
Catheter Precision Inc.'s VIVO System has reached a major milestone.
Company executives said more than 800 procedures have been performed in U.S. and European hospitals using the system, which enables physicians to noninvasively identify an area of ventricular arrhythmia onset.
“We are always looking to the next milestone in clinical usage of our VIVO System and reaching 800 procedures was a goal we set,” Catheter Precision CEO David Jenkins said. “We are gratified by physician adoption so far through our strategic limited launch and look forward to expanding usage in 2023 by growing our sales and clinical support teams to access additional hospitals in new and existing territories. By expanding our footprint, we are bringing a valuable tool to electrophysiologists to help them gain a better understanding of patient anatomy and potentially improve outcomes while providing beneficial healthcare to more patients.”
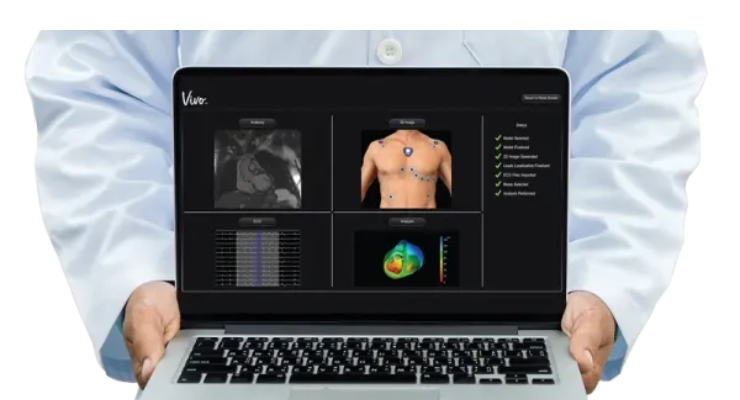
Catheter Precision’s VIVO (View Into Ventricular Onset), is a non-invasive 3D imaging system that enables physicians to identify the origin of arrhythmias pre-procedure, thereby streamlining workflow and reducing procedure time. VIVO has received marketing clearance from the U.S. Food and Drug Administration (FDA) and has been awarded the CE mark. The company also intends to pursue a second generation of Amigo, a previously FDA-cleared and CE mark robotic arm that can be used to remotely control catheters outside of the procedure room. Additionally, Catheter Precision is working toward a commercial launch of a vessel closure device, currently planned for the first half of 2023.
Catheter Precision merged with Ra Medical Systems in early January and is now a wholly-owned subsidiary of the firm.
Company executives said more than 800 procedures have been performed in U.S. and European hospitals using the system, which enables physicians to noninvasively identify an area of ventricular arrhythmia onset.
“We are always looking to the next milestone in clinical usage of our VIVO System and reaching 800 procedures was a goal we set,” Catheter Precision CEO David Jenkins said. “We are gratified by physician adoption so far through our strategic limited launch and look forward to expanding usage in 2023 by growing our sales and clinical support teams to access additional hospitals in new and existing territories. By expanding our footprint, we are bringing a valuable tool to electrophysiologists to help them gain a better understanding of patient anatomy and potentially improve outcomes while providing beneficial healthcare to more patients.”
Catheter Precision’s VIVO (View Into Ventricular Onset), is a non-invasive 3D imaging system that enables physicians to identify the origin of arrhythmias pre-procedure, thereby streamlining workflow and reducing procedure time. VIVO has received marketing clearance from the U.S. Food and Drug Administration (FDA) and has been awarded the CE mark. The company also intends to pursue a second generation of Amigo, a previously FDA-cleared and CE mark robotic arm that can be used to remotely control catheters outside of the procedure room. Additionally, Catheter Precision is working toward a commercial launch of a vessel closure device, currently planned for the first half of 2023.
Catheter Precision merged with Ra Medical Systems in early January and is now a wholly-owned subsidiary of the firm.