Michael Barbella, Managing Editor02.01.23
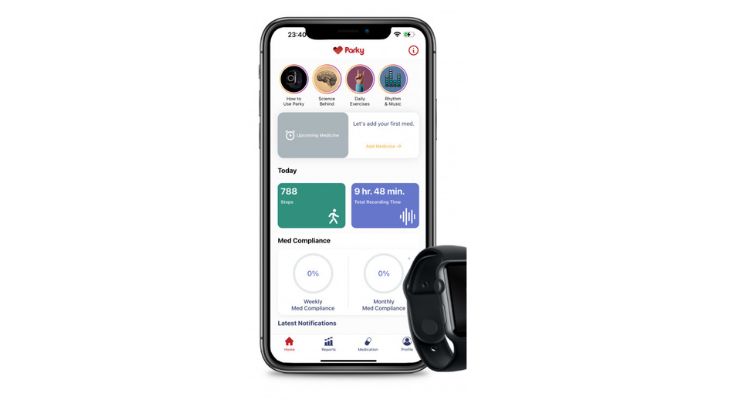
Digital health startup H2o therapeutics has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its prescription mobile app Parky, which monitors Parkinson's Disease. Parky App monitors symptoms such as tremors and dyskinesia in real-time via the use of the Apple Watch. It is a tool for sharing meaningful and reliable data between patients and medical professionals regarding the course of the disease.
"As a woman-founded, non VC-backed company based in Turkey, receiving our first 510(k) clearance is a huge milestone for us. We believe Parky will bring great value to the Parkinson's Disease community as an easily scalable and data-driven product," H2o Founder Yagmur Selin Gulmus said. The company has two other digital therapeutics products in the pipeline based on wearable devices, and FDA submissions are planned to be completed this year.
The app leverages the Movement Disorder API, a tool developed by Apple. The API was validated in a study of 343 participants, including longitudinal tracking of 225 participants for up to six months and used the clinical gold standard (MDS-UPDRS) as a reference. Details of the study were published in Science Translational Medicine in 2021.
Roughly 10 million patients worldwide live with Parkinson's Disease, the fastest-growing neurological disorder. Healthcare professionals in this area need reliable feedback and data for optimized treatment plans. Parky App helps medical professionals to develop a clinical profile of the patient while outside of the clinic. The Parky App strengthens the possibility of data-driven, tailored treatment procedures and bridges the gap between real-life and in-clinic settings.
h2o therapeutics is developing digital therapeutics with a focus on mobile technologies, augmented reality (AR), and artificial intelligence (AI). h2O intends to make real-time human data become a handy tool for disease management in certain therapeutic areas. Its core understanding of digital therapeutics lies in the power of AI, the robustness of its clinical health research, and high levels of user engagement.
"As a woman-founded, non VC-backed company based in Turkey, receiving our first 510(k) clearance is a huge milestone for us. We believe Parky will bring great value to the Parkinson's Disease community as an easily scalable and data-driven product," H2o Founder Yagmur Selin Gulmus said. The company has two other digital therapeutics products in the pipeline based on wearable devices, and FDA submissions are planned to be completed this year.
The app leverages the Movement Disorder API, a tool developed by Apple. The API was validated in a study of 343 participants, including longitudinal tracking of 225 participants for up to six months and used the clinical gold standard (MDS-UPDRS) as a reference. Details of the study were published in Science Translational Medicine in 2021.
Roughly 10 million patients worldwide live with Parkinson's Disease, the fastest-growing neurological disorder. Healthcare professionals in this area need reliable feedback and data for optimized treatment plans. Parky App helps medical professionals to develop a clinical profile of the patient while outside of the clinic. The Parky App strengthens the possibility of data-driven, tailored treatment procedures and bridges the gap between real-life and in-clinic settings.
h2o therapeutics is developing digital therapeutics with a focus on mobile technologies, augmented reality (AR), and artificial intelligence (AI). h2O intends to make real-time human data become a handy tool for disease management in certain therapeutic areas. Its core understanding of digital therapeutics lies in the power of AI, the robustness of its clinical health research, and high levels of user engagement.