Michael Barbella, Managing Editor01.13.23
There's big bucks to be gained in treating healthcare's smallest (youngest) patients.
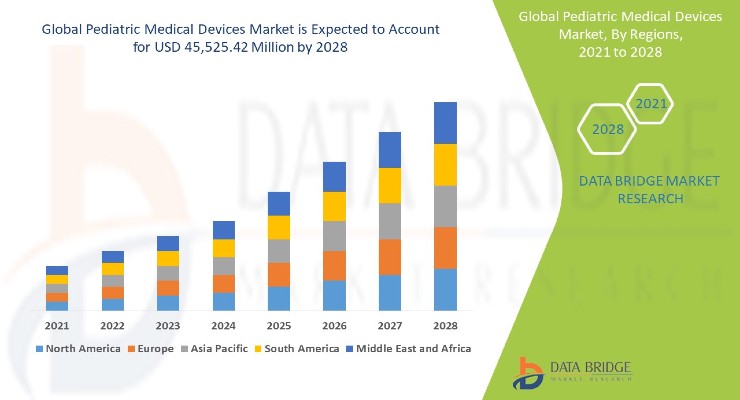
The pediatric medical devices market is expected to swell 7.22% annually over the next six years to reach $45.52 billion by 2028, according to a Data Bridge Market Research report. The increasing prevalence of chronic disease among infants and children will be mainly responsible for the growth.
Pediatric medical devices refer to the equipment or devices used to treat or diagnose diseases and conditions from birth through age 21. Pediatrics is referred to the branch of medicine that deals with the health and medical care of infants, children and adolescents. They are made specifically according to the body structures and functions for the use for children as they are smaller and more active than adults.
The rise in the incidences of chronic disease or infectious disease among the pediatric population such as diphtheria, leukemia, measles, mumps, anemia, asthma, chickenpox, tuberculosis, whooping cough, Lyme disease and pneumonia is one of the major factors driving the market's growth. An increase in demand for cardiology products, neonatal intensive care unit (ICU) devices, and monitoring equipment, and a surge in investments in R&D programs by the government to develop the medical infrastructure in the pediatric field will also help accelerate the sector's expansion. The rise in demand for anesthesia and respiratory devices owning to their effectiveness in treating diseases such as asthma and increasing rate of preterm birth rate across the globe further influences the pediatric medical devices market. Additionally, growing parental awareness programs, increase in investment in R&D activities, and surge in healthcare expenditures will benefit the market as well.
However, the lack of significant biocompatibility and safety criteria for medical devices limit the market;s growth. T
The pediatric medical devices market is segmented on the basis of product and end user. On the basis of product, the market is segmented into cardiology devices, in-vitro diagnostic (IVD) products, diagnostic imaging devices, telemedicine, anesthesia and respiratory care equipment, neonatal ICU devices, and monitoring products. On the basis of end user, the sector is segmented into hospitals, pediatric clinics, ambulatory surgery centers, research and academic institutes, and others.
North America dominates the pediatric medical devices market due to the presence of key players, well-developed healthcare infrastructure, beneficial government initiatives, high prevalence of pediatric chronic diseases, and increase in the number of research partnerships within the region.
Major market players include Abbott, Stryker Corp., Medtronic plc, Boston Scientific Corporation, Cardinal Health, F. Hoffmann-La Roche Ltd, GE, Koninklijke Philips N.V., Baxter, Siemens Healthineers, TSE MEDICAL, DAVID, Hamilton Medical, FRITZ STEPHAN GmbH, Phoenix Medical Systems, Novonate Inc., Elektromag, Trimpeks, Atom Medical Corp., and Johnson & Johnson, among others.
The pediatric medical devices market is expected to swell 7.22% annually over the next six years to reach $45.52 billion by 2028, according to a Data Bridge Market Research report. The increasing prevalence of chronic disease among infants and children will be mainly responsible for the growth.
Pediatric medical devices refer to the equipment or devices used to treat or diagnose diseases and conditions from birth through age 21. Pediatrics is referred to the branch of medicine that deals with the health and medical care of infants, children and adolescents. They are made specifically according to the body structures and functions for the use for children as they are smaller and more active than adults.
The rise in the incidences of chronic disease or infectious disease among the pediatric population such as diphtheria, leukemia, measles, mumps, anemia, asthma, chickenpox, tuberculosis, whooping cough, Lyme disease and pneumonia is one of the major factors driving the market's growth. An increase in demand for cardiology products, neonatal intensive care unit (ICU) devices, and monitoring equipment, and a surge in investments in R&D programs by the government to develop the medical infrastructure in the pediatric field will also help accelerate the sector's expansion. The rise in demand for anesthesia and respiratory devices owning to their effectiveness in treating diseases such as asthma and increasing rate of preterm birth rate across the globe further influences the pediatric medical devices market. Additionally, growing parental awareness programs, increase in investment in R&D activities, and surge in healthcare expenditures will benefit the market as well.
However, the lack of significant biocompatibility and safety criteria for medical devices limit the market;s growth. T
The pediatric medical devices market is segmented on the basis of product and end user. On the basis of product, the market is segmented into cardiology devices, in-vitro diagnostic (IVD) products, diagnostic imaging devices, telemedicine, anesthesia and respiratory care equipment, neonatal ICU devices, and monitoring products. On the basis of end user, the sector is segmented into hospitals, pediatric clinics, ambulatory surgery centers, research and academic institutes, and others.
North America dominates the pediatric medical devices market due to the presence of key players, well-developed healthcare infrastructure, beneficial government initiatives, high prevalence of pediatric chronic diseases, and increase in the number of research partnerships within the region.
Major market players include Abbott, Stryker Corp., Medtronic plc, Boston Scientific Corporation, Cardinal Health, F. Hoffmann-La Roche Ltd, GE, Koninklijke Philips N.V., Baxter, Siemens Healthineers, TSE MEDICAL, DAVID, Hamilton Medical, FRITZ STEPHAN GmbH, Phoenix Medical Systems, Novonate Inc., Elektromag, Trimpeks, Atom Medical Corp., and Johnson & Johnson, among others.