Sam Brusco, Associate Editor11.01.22
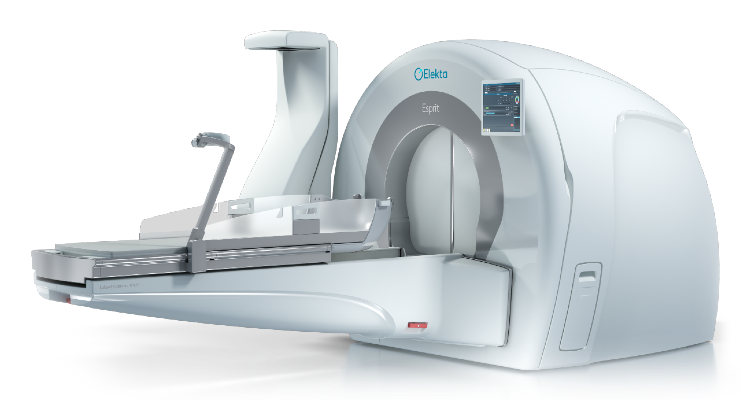
Elekta has earned U.S. Food and Drug Administration (FDA) 510(k) clearance for Elekta Esprit, a new Leksell Gamma Knife radiosurgery platform.
Leksell Gamma Knife aims to be a gentler alternative to open surgery and conventional radiotherapy. It can target the smallest and most challenging intracranial tumors and lesions with minimal effects on healthy tissue, safeguarding motor, sensory, and neurocognitive function.
Verena Schiller, president of Elekta’s Neuroscience Solutions, told the press: “It’s very inspiring to hear how patients treated with Gamma Knife are able to maintain their quality of life and go back to what they love doing after radiosurgery. Thanks to the very low dose delivered outside the target, it’s able to protect surrounding healthy tissue. Esprit takes Gamma Knife to the next level with improvements in both the patient and user experience."
Esprit offers the option of frameless or frame-based workflows as well as remote accessibility and collaboration tools to meet the needs of changing environments.
Elekta Esprit is also CE marked.
Leksell Gamma Knife aims to be a gentler alternative to open surgery and conventional radiotherapy. It can target the smallest and most challenging intracranial tumors and lesions with minimal effects on healthy tissue, safeguarding motor, sensory, and neurocognitive function.
Verena Schiller, president of Elekta’s Neuroscience Solutions, told the press: “It’s very inspiring to hear how patients treated with Gamma Knife are able to maintain their quality of life and go back to what they love doing after radiosurgery. Thanks to the very low dose delivered outside the target, it’s able to protect surrounding healthy tissue. Esprit takes Gamma Knife to the next level with improvements in both the patient and user experience."
Esprit offers the option of frameless or frame-based workflows as well as remote accessibility and collaboration tools to meet the needs of changing environments.
Elekta Esprit is also CE marked.