Sam Brusco, Associate Editor10.25.22
NeuroOne Medical Technologies has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Evo sEEG electrode tech for temporary (less than 30 days) use with recording, monitoring, and stimulation equipment to accomplish all these tasks on electrical signals at the brain’s subsurface level.
"I am extremely proud of the entire NeuroOne team and their relentless pursuit of this clearance. Despite the challenges we faced, our team remained focused and persistent in driving this successful conclusion. This is clearly our most exciting and important accomplishment to date. We are now able to advance our commercialization efforts in partnership with Zimmer Biomet, our distribution and development partner. We look forward to continuing to execute our strategic plan, which next up includes our RF ablation system, the Company's first therapeutic electrode technology," Dave Rosa, CEO of NeuroOne told the press.
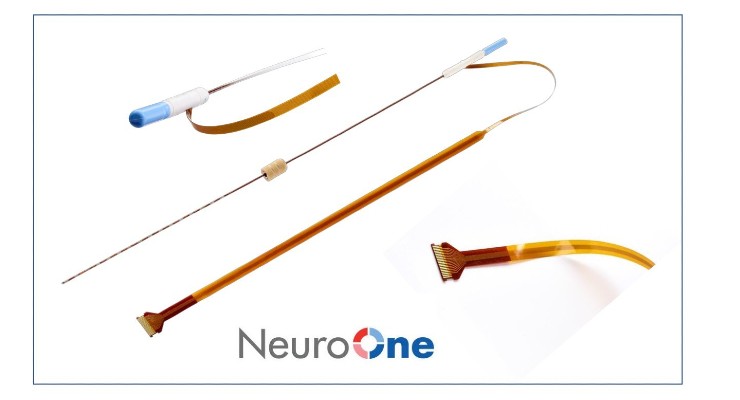
The Evo sEEG system is the company’s second 510(k)-cleared product. Instead of cortical electrodes, sEEG electrodes don’t require removal of the top portion of the skull. Potential advantages include higher signal clarity and reduced noise, better tactile feedback during insertion, and faster order fulfillment.
"I am extremely proud of the entire NeuroOne team and their relentless pursuit of this clearance. Despite the challenges we faced, our team remained focused and persistent in driving this successful conclusion. This is clearly our most exciting and important accomplishment to date. We are now able to advance our commercialization efforts in partnership with Zimmer Biomet, our distribution and development partner. We look forward to continuing to execute our strategic plan, which next up includes our RF ablation system, the Company's first therapeutic electrode technology," Dave Rosa, CEO of NeuroOne told the press.
The Evo sEEG system is the company’s second 510(k)-cleared product. Instead of cortical electrodes, sEEG electrodes don’t require removal of the top portion of the skull. Potential advantages include higher signal clarity and reduced noise, better tactile feedback during insertion, and faster order fulfillment.