Sam Brusco, Associate Editor09.09.22
Yesterday, the U.S. Food and Drug Administration (FDA) warned healthcare providers about potential MitraClip clip delivery system malfunctions in the device’s clip lock.
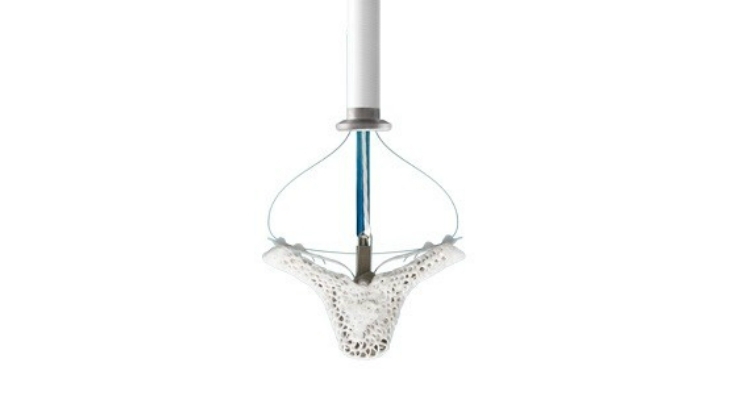
MitraClip is used to repair the mitral heart valve to treat mitral regurgitation (MR). According to MitraClip maker Abbott Labs, it’s the only percutaneous repair device approved in the U.S. to treat MR patients.
Abbott reported a higher rate of clip lock malfunctions observed before and after deployment of the clip. The events occurred in about 1.3% of MitraClip procedures and affected all device models.
Patient risks of a clip lock malfunction include ineffective mitral regurgitation treatment and need for further interventions, which can lead to potential bleeding, implantation complications from additional clips, and longer surgery times.
According to the FDA, most of the reported clip lock malfunctions haven’t resulted in adverse outcomes to patients. The FDA believes that MitraClip’s probable benefits outweigh probable risks based on the data from malfunctions and associated risks.
Abbott’s correction letter includes recommendations to minimize the risk of clip lock malfunction: guidance for implant positioning, locking sequences, establishing clip arm angle, preparation for clip release, and avoiding excessive force and manipulation when unlocking the clip during device preparation and during the procedure.
MitraClip is used to repair the mitral heart valve to treat mitral regurgitation (MR). According to MitraClip maker Abbott Labs, it’s the only percutaneous repair device approved in the U.S. to treat MR patients.
Abbott reported a higher rate of clip lock malfunctions observed before and after deployment of the clip. The events occurred in about 1.3% of MitraClip procedures and affected all device models.
Patient risks of a clip lock malfunction include ineffective mitral regurgitation treatment and need for further interventions, which can lead to potential bleeding, implantation complications from additional clips, and longer surgery times.
According to the FDA, most of the reported clip lock malfunctions haven’t resulted in adverse outcomes to patients. The FDA believes that MitraClip’s probable benefits outweigh probable risks based on the data from malfunctions and associated risks.
Abbott’s correction letter includes recommendations to minimize the risk of clip lock malfunction: guidance for implant positioning, locking sequences, establishing clip arm angle, preparation for clip release, and avoiding excessive force and manipulation when unlocking the clip during device preparation and during the procedure.