Sam Brusco, Associate Editor06.01.22
The U.S. Food and Drug Administration (FDA) has identified Abbott’s recall of its Dragonfly OpStar imaging catheter as Class I due to the potential for a loose catheter marker band that may cause harm.
Abbott began recalling the devices April 11. There are 4,800 devices affected by this recall.
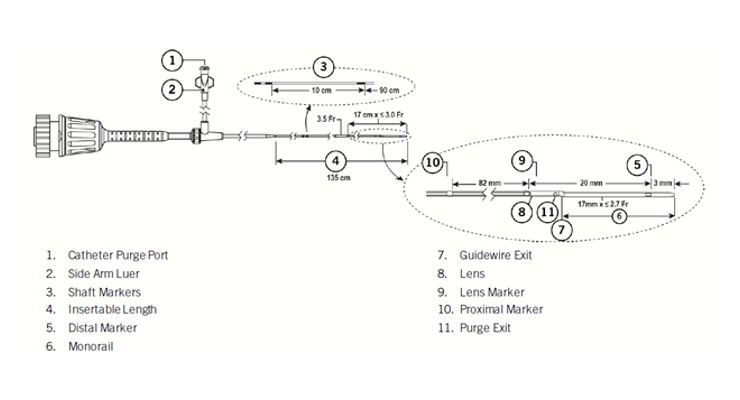
Dragonfly OpStar is paired with optical coherence tomography (OCT) to image coronary arteries in people who are candidates for catheter-based, minimally invasive, interventional procedures for coronary artery disease.
Abbott recalled the imaging catheters because the proximal marker band may loosen and, in two instances, has separated from the catheter when being used on a person.
A loose market separating from the device could remain in the body after the catheter is removed, risking vascular injuries like embolism, thrombosis, dissection, ischemia, infarction, infection, or death.
Five incidents and one injury have thus far been reported due to this issue, but no deaths have yet been associated.
Abbott urged customers to immediately stop using Dragonfly OpStar and return unused devices to the company.
Abbott began recalling the devices April 11. There are 4,800 devices affected by this recall.
Dragonfly OpStar is paired with optical coherence tomography (OCT) to image coronary arteries in people who are candidates for catheter-based, minimally invasive, interventional procedures for coronary artery disease.
Abbott recalled the imaging catheters because the proximal marker band may loosen and, in two instances, has separated from the catheter when being used on a person.
A loose market separating from the device could remain in the body after the catheter is removed, risking vascular injuries like embolism, thrombosis, dissection, ischemia, infarction, infection, or death.
Five incidents and one injury have thus far been reported due to this issue, but no deaths have yet been associated.
Abbott urged customers to immediately stop using Dragonfly OpStar and return unused devices to the company.