Sam Brusco, Associate Editor01.18.22
Orthodontic software firm SoftSmile received U.S. Food and Drug Administration (FDA) 510(k) clearance for its VISION software.
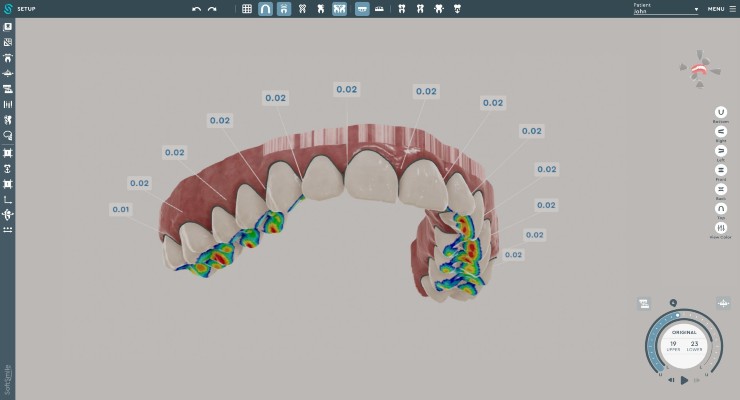
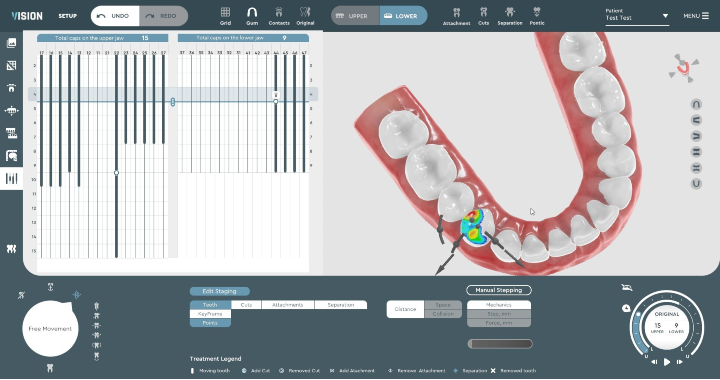
VISION is an end-to-end treatment planning software made for doctors by doctors. It enables creation of precise aligner treatment plans, which are then made using 3D printing technologies.
The company is currently working with aligner manufacturers. VISION’s capabilities include automated segmentation as well as intuitive and comprehensive staging. The software automatically generates key features of a digitally optimized treatment plan. It can be used without having to utilize third-party designers and/or manufacturers.
"FDA clearance is a huge step for SoftSmile because it establishes Vision's position as a leading software service that complies with all requirements imposed by FDA and is safe for use in the medical practice,” Khamzat Asabaev, SoftSmile founder and CEO told the press. “There are just a few orthodontic treatment planners that have obtained FDA clearance. SoftSmile's new status is a groundbreaking assurance for the market because the FDA is the best quality check for doctors and patients. This achievement opens new opportunities for the company and proves that our team can solve any type of challenge. It took nearly 18 months, enormous resources, and the coordinated efforts of our engineers and lawyers to be granted FDA clearance. Fortunately, we have only the best at SoftSmile which has now been given the FDA's stamp of approval.”
VISION is an end-to-end treatment planning software made for doctors by doctors. It enables creation of precise aligner treatment plans, which are then made using 3D printing technologies.
The company is currently working with aligner manufacturers. VISION’s capabilities include automated segmentation as well as intuitive and comprehensive staging. The software automatically generates key features of a digitally optimized treatment plan. It can be used without having to utilize third-party designers and/or manufacturers.

"FDA clearance is a huge step for SoftSmile because it establishes Vision's position as a leading software service that complies with all requirements imposed by FDA and is safe for use in the medical practice,” Khamzat Asabaev, SoftSmile founder and CEO told the press. “There are just a few orthodontic treatment planners that have obtained FDA clearance. SoftSmile's new status is a groundbreaking assurance for the market because the FDA is the best quality check for doctors and patients. This achievement opens new opportunities for the company and proves that our team can solve any type of challenge. It took nearly 18 months, enormous resources, and the coordinated efforts of our engineers and lawyers to be granted FDA clearance. Fortunately, we have only the best at SoftSmile which has now been given the FDA's stamp of approval.”