Business Wire10.15.21
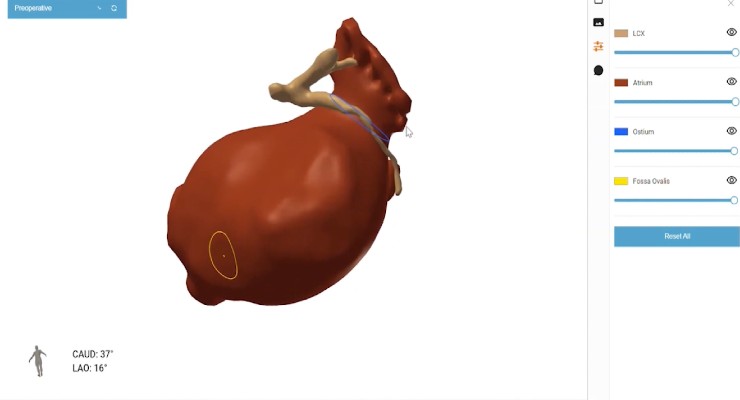
FEops received De Novo authorization from the U.S. Food and Drug Administration (FDA) for FEops HEARTguide pre-operative planning of left atrial appendage occlusion with the Boston Scientific WATCHMAN device*. FEops HEARTguide will enable physicians to virtually model clinical scenarios with different implant positions and sizes of the WATCHMAN device helping the heart team to select the optimal size and position for every individual patient.
“The pre-operative insight provided by FEops HEARTguide is powerful and can help me to optimize decision making for selecting optimal device size and position," said Dr. Jacqueline Saw, Vancouver General Hospital, Canada. “FEops HEARTguide is an intuitive platform I can use to discuss cases with the entire heart team to streamline my pre-op planning workflow.”
“FDA authorization of FEops HEARTguide is a significant milestone as this is the first Interventional Cardiovascular Implant Simulation Software Device cleared on the US market,” said Peter Mortier, Ph.D., co-founder and CTO of FEops. “Research on this technology began more than 10 years ago. We have continued to develop it with the goal of helping healthcare professionals identify the most appropriate treatment strategy for each patient precisely, safely and efficiently. This De Novo clearance is only a first step, and we are already preparing for FDA 510(k) submissions for FEops HEARTguide LAAo workflow with Abbott’s Amplatzer Amulet device and Boston Scientific’s WATCHMAN FLX device.”
FEops HEARTguide is a one-in-its-kind procedure planning platform for structural heart interventions that provides physicians with unique insights to evaluate device sizing and positioning pre-operatively. The platform uses digital twin technology based on patient-specific virtual replicas of the heart. Besides the recent FDA clearance to enter the US market with its LAAo workflow with WATCHMAN, FEops HEARTguide is also commercially available in the European Union, UK, Canada, and Australia for its TAVI and LAAo workflows.
*The Owner/Operator Number for this Registration is: 10082838 and the listing number: DEN200030.
“The pre-operative insight provided by FEops HEARTguide is powerful and can help me to optimize decision making for selecting optimal device size and position," said Dr. Jacqueline Saw, Vancouver General Hospital, Canada. “FEops HEARTguide is an intuitive platform I can use to discuss cases with the entire heart team to streamline my pre-op planning workflow.”
“FDA authorization of FEops HEARTguide is a significant milestone as this is the first Interventional Cardiovascular Implant Simulation Software Device cleared on the US market,” said Peter Mortier, Ph.D., co-founder and CTO of FEops. “Research on this technology began more than 10 years ago. We have continued to develop it with the goal of helping healthcare professionals identify the most appropriate treatment strategy for each patient precisely, safely and efficiently. This De Novo clearance is only a first step, and we are already preparing for FDA 510(k) submissions for FEops HEARTguide LAAo workflow with Abbott’s Amplatzer Amulet device and Boston Scientific’s WATCHMAN FLX device.”
FEops HEARTguide is a one-in-its-kind procedure planning platform for structural heart interventions that provides physicians with unique insights to evaluate device sizing and positioning pre-operatively. The platform uses digital twin technology based on patient-specific virtual replicas of the heart. Besides the recent FDA clearance to enter the US market with its LAAo workflow with WATCHMAN, FEops HEARTguide is also commercially available in the European Union, UK, Canada, and Australia for its TAVI and LAAo workflows.
*The Owner/Operator Number for this Registration is: 10082838 and the listing number: DEN200030.