Thermo Fisher Scientific05.15.20
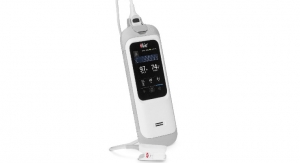
Thermo Fisher Scientific, the world leader in serving science, announced that its Thermo Scientific Cascadion SM Clinical Analyzer is now commercially available in the United States, with the Cascadion SM 25-Hydroxy Vitamin D assay. Designed to deliver accurate, traceable measurements of vitamin D2 (ergocalciferol) and D3 (cholecalciferol), this serum assay reports total vitamin D results alongside its components. Clinical laboratories can now benefit from a complete system bringing the accuracy, sensitivity and specificity benefits of Thermo Fisher’s gold standard liquid chromatography-tandem mass spectrometry (LC-MS/MS) technology into routine clinical workflows.
Incorporating sample processing, LC separation and MS detection capabilities in a single solution, the Cascadion system is fully automated and enables load-up and walkaway sample management, eliminating the need for multiple connected components. The system offers end-to-end automation that is easy to use by any qualified laboratory personnel with minimal training, thus streamlining and optimizing the analytical process.
The Cascadion system analyzes samples directly from qualified primary blood collection tubes, while the Cascadion SM 25-Hydroxy Vitamin D Assay incorporates fully barcoded components, all traceable to a specific result. The vitamin D calibrators and controls are traceable according to the National Institute of Standards and Technology (NIST) and assay calibration remains stable for up to 30 days.
"The Centers for Disease Control and Prevention has established LC-MS/MS as the reference method for vitamin D testing," explains Bill Östman, vice president and general manager, Thermo Fisher Scientific. "However, LC-MS/MS has traditionally been a sophisticated technology, requiring specialized knowledge and significant hands-on time for method development. We developed the Cascadion system and the ready-to-use, pre-validated 25-Hydroxy Vitamin D assay to simplify and accelerate the implementation of LC-MS/MS in clinical settings without the need for extensive technical expertise."
The Cascadion analyzer is listed with the U.S. Food and Drug Administration (FDA) as a Class I instrument, while the Cascadion SM 25-Hydroxy Vitamin D assay is FDA-listed as Class II exempt with special controls.
Incorporating sample processing, LC separation and MS detection capabilities in a single solution, the Cascadion system is fully automated and enables load-up and walkaway sample management, eliminating the need for multiple connected components. The system offers end-to-end automation that is easy to use by any qualified laboratory personnel with minimal training, thus streamlining and optimizing the analytical process.
The Cascadion system analyzes samples directly from qualified primary blood collection tubes, while the Cascadion SM 25-Hydroxy Vitamin D Assay incorporates fully barcoded components, all traceable to a specific result. The vitamin D calibrators and controls are traceable according to the National Institute of Standards and Technology (NIST) and assay calibration remains stable for up to 30 days.
"The Centers for Disease Control and Prevention has established LC-MS/MS as the reference method for vitamin D testing," explains Bill Östman, vice president and general manager, Thermo Fisher Scientific. "However, LC-MS/MS has traditionally been a sophisticated technology, requiring specialized knowledge and significant hands-on time for method development. We developed the Cascadion system and the ready-to-use, pre-validated 25-Hydroxy Vitamin D assay to simplify and accelerate the implementation of LC-MS/MS in clinical settings without the need for extensive technical expertise."
The Cascadion analyzer is listed with the U.S. Food and Drug Administration (FDA) as a Class I instrument, while the Cascadion SM 25-Hydroxy Vitamin D assay is FDA-listed as Class II exempt with special controls.