Dianosic05.15.20
Dianosic SAS, a Strasbourg-based startup specializing in innovative solutions for ear, nose, and throat (ENT) conditions, announced that it has obtained CE marking for CAVI-T, a low-pressure asymmetric balloon for treating spontaneous and post-operative intranasal bleeding. This class IIa certification will enable the company to begin marketing the device in the European Union.
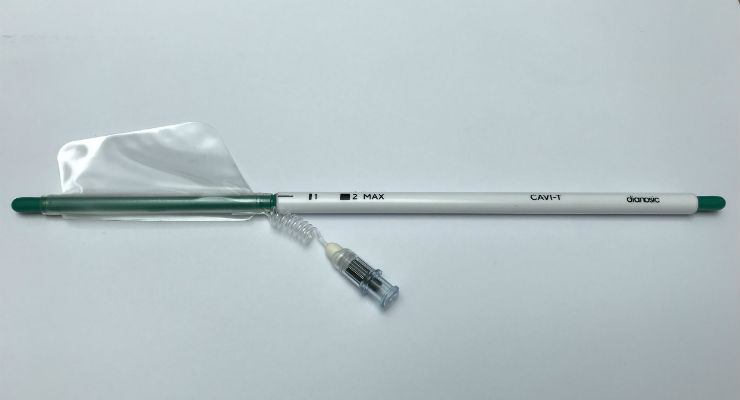
CAVI-T, which is designed to treat intranasal bleeding (epistaxis), is patented in France and will be soon in Europe, the United States, Japan and China. It is the first asymmetric intranasal balloon that stops bleeding by applying low-force compression while adapting to the anatomy of the nasal fossa. The balloon can remain in place for one to three days.
At present, this atraumatic, easy-to-use medical device is aimed primarily at hospitals. A pilot study between April and June 2019, led by Professor Christian Debry, head of the ENT department at Strasbourg University Hospital, demonstrated its safety and efficacy. The balloon was effective in stopping bleeding in 90 percent of patients and significantly improved their quality of life. This feasibility study also demonstrated the good safety profile of CAVI-T, with a mean pain score on product insertion and removal of 4.7 and 1.6 respectively. (Evaluated on a Visual Analogue Scale ranging from one to 10).
“Obtaining CE marking is an important recognition of Dianosic’s expertise and the ability of its team to conceive and develop innovative products to treat ENT disorders,” said Marc Augustin, president of Dianosic.
“Following this major step in our development, we are ready to prepare the commercial launch of the product in Europe, which is expected to take place during the second quarter of 2020. Dianosic’s entry into the market represents a prime opportunity for growth, with the potential to reach close to five million patients across Europe and the United States,” said Philippe Bastide, CEO of Dianosic.
CAVI-T, which is designed to treat intranasal bleeding (epistaxis), is patented in France and will be soon in Europe, the United States, Japan and China. It is the first asymmetric intranasal balloon that stops bleeding by applying low-force compression while adapting to the anatomy of the nasal fossa. The balloon can remain in place for one to three days.
At present, this atraumatic, easy-to-use medical device is aimed primarily at hospitals. A pilot study between April and June 2019, led by Professor Christian Debry, head of the ENT department at Strasbourg University Hospital, demonstrated its safety and efficacy. The balloon was effective in stopping bleeding in 90 percent of patients and significantly improved their quality of life. This feasibility study also demonstrated the good safety profile of CAVI-T, with a mean pain score on product insertion and removal of 4.7 and 1.6 respectively. (Evaluated on a Visual Analogue Scale ranging from one to 10).
“Obtaining CE marking is an important recognition of Dianosic’s expertise and the ability of its team to conceive and develop innovative products to treat ENT disorders,” said Marc Augustin, president of Dianosic.
“Following this major step in our development, we are ready to prepare the commercial launch of the product in Europe, which is expected to take place during the second quarter of 2020. Dianosic’s entry into the market represents a prime opportunity for growth, with the potential to reach close to five million patients across Europe and the United States,” said Philippe Bastide, CEO of Dianosic.