Spectrum Solutions LLC04.03.20
Spectrum DNA, a division of Salt Lake City-based Spectrum Solutions LLC, today announced that using the Spectrum DNA SDNA-1000 Whole Saliva Collection Device, researchers from RUCDR Infinite Biologics at Rutgers University have successfully validated saliva as being a viable biosample source for COVID-19 detection when compared to nasopharyngeal or oropharyngeal swabs. The resourceful discovery has come just in time to answer the consistent exponential increase in test demand.
“This is it. This is what makes America so great,” Said Bill Phillips, Spectrum’s chief operating officer. “Challenge and adversity throw up roadblocks and we partner together to discover, and deliver, not only an answer to the supply shortage for sample collection but uncover a solution that is actually better than what we were using in the first place.”
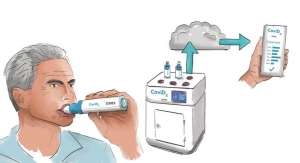
Saliva testing is a new development that under a medical provider’s direction, allows for simple self-administered sample collection that even those in quarantine or self-isolation can use. Researchers at Rutgers’s RUCDR Infinite Biologics compared swab-collected biosamples head-to-head against saliva biosamples using the Spectrum DNA whole saliva collection device and its patented blue preservation solution. Saliva not only demonstrated to be a robust source of viral RNA, but when preserved with Spectrum’s patented preservation solution it was seen to protect COVID-19 transcripts for an extended period of time making sample collection and transport to CLIA testing labs more efficient and amenable to global testing. Adding in the capability for self-collection uncovers additional opportunity to protect medical teams helping to reduce the risk of further exposure and spread, opposed to nasal swab biosample collection requiring up close medical assistance.
“It had been previously reported on how saliva was being successfully used for viral DNA/RNA extraction. Due to this state of emergency and medical supply shortage we decided to test it for ourselves using Spectrum saliva collection devices. Our side-by-side test comparison of over 75 patient samples demonstrated that the use of saliva to extract viral RNA was in fact a robust source for COVID-19 detection and equal in performance to the approved swab-based collection samples,” said Andrew Brooks, Ph.D. chief operating officer, RUCDR Infinite Biologics and Professor of Genetics at Rutgers University. “In addition, the preservation solution from the Spectrum whole saliva collection device preserved the COVID-19 viral RNA for testing enabling for easier logistics from sample collection to the testing laboratory.”
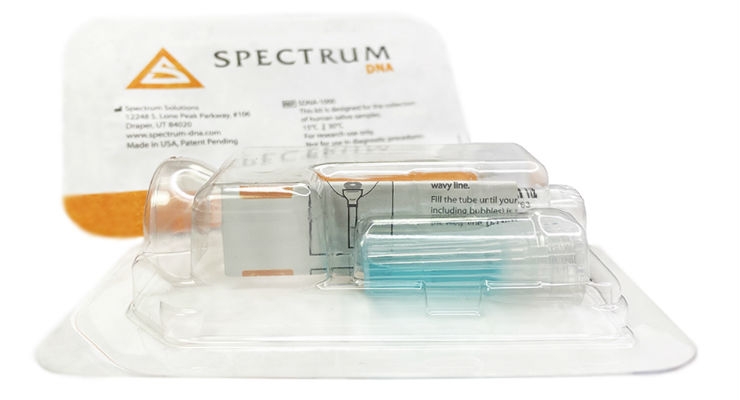
RUCDR is currently testing for COVID-19 using the Spectrum DNA SDNA-1000 whole saliva collection device leveraging two easily accessible COVID-19 detection assays under emergency use authorization from the U.S. Food and Drug Administration. Any laboratory with the ability to test for COVID-19 and the willingness to contribute their assistance during this state of emergency can contact Spectrum for access to saliva testing support documentation, approved operating procedures and protocols required to file an EUA. Contact Spectrum DNA by email or 801-569-0465. Information on the SDNA-1000 saliva collection device can be found here.
“As demand for COVID-19 testing continues to dramatically climb, supplies for many of the critical components are just not keeping pace. The discovery that saliva is a viable and more convenient source of biomaterial for testing will immediately help hospital networks and labs bypassing the critical shortage of supplies including nasal and throat swabs for COVID-19 testing,” said Stephen J. Fanning, CEO of Spectrum Solutions. "A saliva test will also allow for a broad and more expansive population screening for the virus than what has been possible to date during this global health crisis.”
“These exciting results have a tremendous impact on testing. We encourage any clinical laboratory or reference laboratory wanting to validate our protocols and submit for their EUA testing approval to contact Spectrum for guidance. The more of us approved for testing, the faster we can deliver testing relief to those desperately waiting for answers,” said Dr. Brooks.
Spectrum Solutions has manufactured more than 10 million Saliva Collection devices for distribution worldwide. The SDNA-1000 Saliva Collection Device is Spectrum’s latest device, which will help to lead the saliva collection market in molecular and other diagnostic applications. This fit-for-purpose and technically superior whole saliva collection device has been engineered to reduce self-collection error, is bacteriostatic, and delivers the consistent high-quality, high-yield samples for any medical diagnostic or clinical research required application. The patented preservation solution stabilizes viral samples within the saliva matrix effectively reducing additional exposure-risks to health professionals. As a result, ensuring sample stability at a variety of temperatures enable the primary sample to be shipped as well as easily stored in case repeat testing is required.
The product is available now and currently being used as a biosample collection option for viral DNA/RNA COVID-19 testing--allowing hospital networks, health departments, and clinical research organizations to accept tens of thousands of samples per day for SARS-CoV-2 coronavirus testing and analysis. Spectrum does its product manufacturing of the SDNA-1000 saliva collection device in-house at their facility in Salt Lake City with the capacity, and proven scalability to meet the rapid growing need for COVID-19 testing devices.
“This is it. This is what makes America so great,” Said Bill Phillips, Spectrum’s chief operating officer. “Challenge and adversity throw up roadblocks and we partner together to discover, and deliver, not only an answer to the supply shortage for sample collection but uncover a solution that is actually better than what we were using in the first place.”
Saliva testing is a new development that under a medical provider’s direction, allows for simple self-administered sample collection that even those in quarantine or self-isolation can use. Researchers at Rutgers’s RUCDR Infinite Biologics compared swab-collected biosamples head-to-head against saliva biosamples using the Spectrum DNA whole saliva collection device and its patented blue preservation solution. Saliva not only demonstrated to be a robust source of viral RNA, but when preserved with Spectrum’s patented preservation solution it was seen to protect COVID-19 transcripts for an extended period of time making sample collection and transport to CLIA testing labs more efficient and amenable to global testing. Adding in the capability for self-collection uncovers additional opportunity to protect medical teams helping to reduce the risk of further exposure and spread, opposed to nasal swab biosample collection requiring up close medical assistance.
“It had been previously reported on how saliva was being successfully used for viral DNA/RNA extraction. Due to this state of emergency and medical supply shortage we decided to test it for ourselves using Spectrum saliva collection devices. Our side-by-side test comparison of over 75 patient samples demonstrated that the use of saliva to extract viral RNA was in fact a robust source for COVID-19 detection and equal in performance to the approved swab-based collection samples,” said Andrew Brooks, Ph.D. chief operating officer, RUCDR Infinite Biologics and Professor of Genetics at Rutgers University. “In addition, the preservation solution from the Spectrum whole saliva collection device preserved the COVID-19 viral RNA for testing enabling for easier logistics from sample collection to the testing laboratory.”
RUCDR is currently testing for COVID-19 using the Spectrum DNA SDNA-1000 whole saliva collection device leveraging two easily accessible COVID-19 detection assays under emergency use authorization from the U.S. Food and Drug Administration. Any laboratory with the ability to test for COVID-19 and the willingness to contribute their assistance during this state of emergency can contact Spectrum for access to saliva testing support documentation, approved operating procedures and protocols required to file an EUA. Contact Spectrum DNA by email or 801-569-0465. Information on the SDNA-1000 saliva collection device can be found here.
“As demand for COVID-19 testing continues to dramatically climb, supplies for many of the critical components are just not keeping pace. The discovery that saliva is a viable and more convenient source of biomaterial for testing will immediately help hospital networks and labs bypassing the critical shortage of supplies including nasal and throat swabs for COVID-19 testing,” said Stephen J. Fanning, CEO of Spectrum Solutions. "A saliva test will also allow for a broad and more expansive population screening for the virus than what has been possible to date during this global health crisis.”
“These exciting results have a tremendous impact on testing. We encourage any clinical laboratory or reference laboratory wanting to validate our protocols and submit for their EUA testing approval to contact Spectrum for guidance. The more of us approved for testing, the faster we can deliver testing relief to those desperately waiting for answers,” said Dr. Brooks.
Spectrum Solutions has manufactured more than 10 million Saliva Collection devices for distribution worldwide. The SDNA-1000 Saliva Collection Device is Spectrum’s latest device, which will help to lead the saliva collection market in molecular and other diagnostic applications. This fit-for-purpose and technically superior whole saliva collection device has been engineered to reduce self-collection error, is bacteriostatic, and delivers the consistent high-quality, high-yield samples for any medical diagnostic or clinical research required application. The patented preservation solution stabilizes viral samples within the saliva matrix effectively reducing additional exposure-risks to health professionals. As a result, ensuring sample stability at a variety of temperatures enable the primary sample to be shipped as well as easily stored in case repeat testing is required.
The product is available now and currently being used as a biosample collection option for viral DNA/RNA COVID-19 testing--allowing hospital networks, health departments, and clinical research organizations to accept tens of thousands of samples per day for SARS-CoV-2 coronavirus testing and analysis. Spectrum does its product manufacturing of the SDNA-1000 saliva collection device in-house at their facility in Salt Lake City with the capacity, and proven scalability to meet the rapid growing need for COVID-19 testing devices.