Globe Newswire10.18.19
Medtronic plc, launched the GI Genius intelligent endoscopy module yesterday at UEG Week in Barcelona, Spain. The GI Genius module uses artificial intelligence to detect colorectal polyps, providing physicians with a powerful new solution in the fight against colorectal cancer.
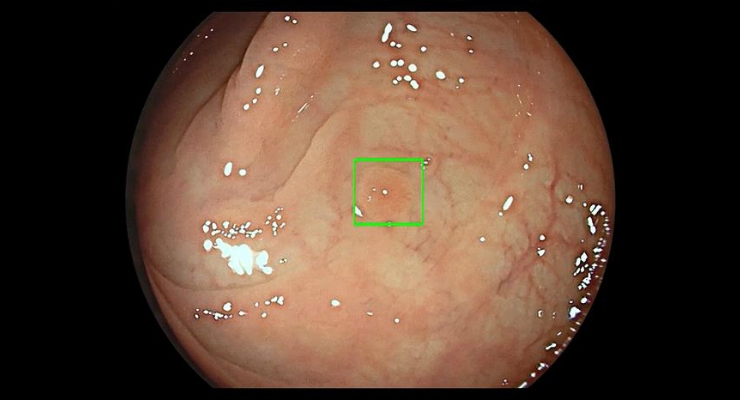
The GI Genius module uses advanced artificial intelligence to highlight the presence of pre-cancerous lesions with a visual marker in real-time—serving as an ever vigilant second observer. Studies have shown that having a second observer can increase polyp detection rates; every 1 percent increase in adenoma detection rate reduces the risk of colorectal cancer by 3 percent.1 Colorectal cancer (CRC) is the third most common form of cancer globally with 1.8 million new cases every year.2
"One of the key factors to maximizing the prevention of colorectal cancer is the integration of advanced technologies like artificial intelligence into daily practice. The use of artificial intelligence in gastroenterology ushers in a new era of diagnostic endoscopy that can improve the quality of colonoscopies. In my experience, the GI Genius module can be extremely precise in identifying lesions in the colonic mucosa that can be difficult to detect and may have been missed. This change in daily practice has the potential to improve diagnosis and overall outcomes for patients that may have colorectal cancer," said Prof. Alessandro Repici, head of gastroenterology at Humanitas Hospital in Rozzano, Milan.
"Medtronic is focused on preventing colorectal cancer by early detection of pre-cancerous polyps with AI-assisted technologies," said Giovanni Di Napoli, vice president and general manager of the Gastrointestinal & Hepatology business, which is part of the Minimally Invasive Therapies Group at Medtronic. "The GI Genius module automatically detects polyps, including small flat polyps that may go undetected thus increasing accuracy and reducing the risk of interval cancers which can occur between colonoscopies."
To bring this technology to market, Medtronic entered a worldwide distribution agreement with Cosmo Pharmaceuticals, the sole manufacturer of the artificial intelligent software and device. GI Genius intelligent endoscopy module is CE-marked and is available in select European markets. The module does not have FDA clearance and therefore is not available for sale in the United States.
References:
1Corley DA, Jenson CD, Marks AR JR, et al. NEJM 2014;370:1298-306.
2 http://www.crcprevention.eu/index.php?pg=colorectal-cancer-epidemiology
The GI Genius module uses advanced artificial intelligence to highlight the presence of pre-cancerous lesions with a visual marker in real-time—serving as an ever vigilant second observer. Studies have shown that having a second observer can increase polyp detection rates; every 1 percent increase in adenoma detection rate reduces the risk of colorectal cancer by 3 percent.1 Colorectal cancer (CRC) is the third most common form of cancer globally with 1.8 million new cases every year.2
"One of the key factors to maximizing the prevention of colorectal cancer is the integration of advanced technologies like artificial intelligence into daily practice. The use of artificial intelligence in gastroenterology ushers in a new era of diagnostic endoscopy that can improve the quality of colonoscopies. In my experience, the GI Genius module can be extremely precise in identifying lesions in the colonic mucosa that can be difficult to detect and may have been missed. This change in daily practice has the potential to improve diagnosis and overall outcomes for patients that may have colorectal cancer," said Prof. Alessandro Repici, head of gastroenterology at Humanitas Hospital in Rozzano, Milan.
"Medtronic is focused on preventing colorectal cancer by early detection of pre-cancerous polyps with AI-assisted technologies," said Giovanni Di Napoli, vice president and general manager of the Gastrointestinal & Hepatology business, which is part of the Minimally Invasive Therapies Group at Medtronic. "The GI Genius module automatically detects polyps, including small flat polyps that may go undetected thus increasing accuracy and reducing the risk of interval cancers which can occur between colonoscopies."
To bring this technology to market, Medtronic entered a worldwide distribution agreement with Cosmo Pharmaceuticals, the sole manufacturer of the artificial intelligent software and device. GI Genius intelligent endoscopy module is CE-marked and is available in select European markets. The module does not have FDA clearance and therefore is not available for sale in the United States.
References:
1Corley DA, Jenson CD, Marks AR JR, et al. NEJM 2014;370:1298-306.
2 http://www.crcprevention.eu/index.php?pg=colorectal-cancer-epidemiology