Business Wire10.22.18
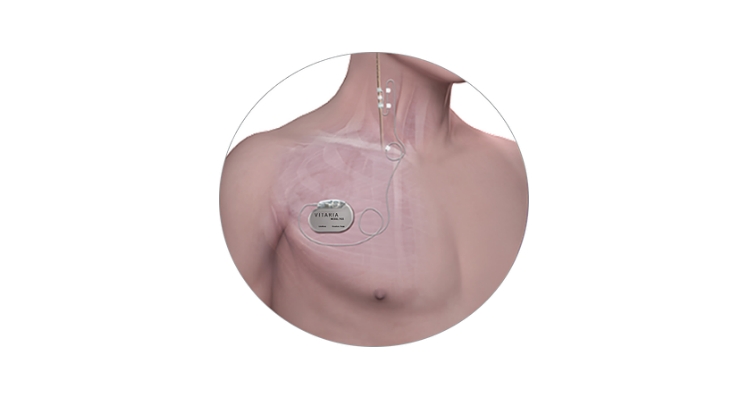
LivaNova PLC announced the first successful implantation of the VITARIA System in a patient enrolled in the Autonomic Regulation Therapy to Enhance Myocardial Function and Reduce Progression of Heart Failure With Reduced Ejection Fraction (ANTHEM-HFrEF) Pivotal Study. The study is an international, multi-center, randomized trial to evaluate the VITARIA System for the treatment of advanced heart failure. The first implant in the pivotal study was completed at UnityPoint Health – St. Luke’s Hospital in Cedar Rapids, Iowa, by Dr. Jared Kray.
“Patients with advanced heart failure continue to need additional and complementary therapy options, since drug treatment alone has slowed but not stopped heart failure progression,” said Dr. Ron M. Oren, heart failure cardiologist and lead investigator for the study at the Heart and Vascular Institute in Cedar Rapids. “We are pleased to participate in this important study to critically evaluate the safety and efficacy of Autonomic Regulation Therapy delivered by the VITARIA System.”
The LivaNova VITARIA System delivers Autonomic Regulation Therapy (ART) using Vagus Nerve Stimulation (VNS) in patients who continue to experience symptoms of heart failure despite receiving guideline-directed medical therapy. The VITARIA System received CE Mark approval in 2015 and received expedited access pathway designation as a breakthrough technology from the U.S. Food and Drug Administration in 2017. The therapy may be the first implantable active neurostimulation system for the treatment of advanced heart failure, if the study demonstrates statistically and clinically meaningful improvements in pre-specified endpoints related to heart failure symptom improvement, hospitalization and survival.
“LivaNova is dedicated to changing lives through the development and introduction of novel technologies, which address large unmet needs in the global medical community. Autonomic Regulation Therapy is based on our experience derived from VNS Therapy, which is used to treat other disorders,” said Edward Andrle, LivaNova general manager of Neuromodulation. “The ANTHEM-HFrEF Pivotal Study brings us closer to offering the VITARIA System to a large segment of the more than 20 million people currently battling heart failure around the world.”
“Autonomic Regulation Therapy holds promise for patients with advanced heart failure, and we are pleased that this landmark study is underway,” said Dr. Marvin Konstam, principal investigator for the ANTHEM-HFrEF Pivotal Study and chief physician of the CardioVascular Center at Tufts Medical Center in Boston, Mass.
LivaNova PLC is a global medical technology company built on nearly five decades of experience and a relentless commitment to improve the lives of patients around the world. LivaNova’s advanced technologies and breakthrough treatments provide meaningful solutions for the benefit of patients, healthcare professionals and healthcare systems. Headquartered in London, LivaNova has a presence in more than 100 countries worldwide. The company currently employs approximately 4,000 employees. LivaNova operates as two businesses: Cardiac Surgery and Neuromodulation, with operating headquarters in Mirandola, Italy, and Houston, Texas, respectively.
“Patients with advanced heart failure continue to need additional and complementary therapy options, since drug treatment alone has slowed but not stopped heart failure progression,” said Dr. Ron M. Oren, heart failure cardiologist and lead investigator for the study at the Heart and Vascular Institute in Cedar Rapids. “We are pleased to participate in this important study to critically evaluate the safety and efficacy of Autonomic Regulation Therapy delivered by the VITARIA System.”
The LivaNova VITARIA System delivers Autonomic Regulation Therapy (ART) using Vagus Nerve Stimulation (VNS) in patients who continue to experience symptoms of heart failure despite receiving guideline-directed medical therapy. The VITARIA System received CE Mark approval in 2015 and received expedited access pathway designation as a breakthrough technology from the U.S. Food and Drug Administration in 2017. The therapy may be the first implantable active neurostimulation system for the treatment of advanced heart failure, if the study demonstrates statistically and clinically meaningful improvements in pre-specified endpoints related to heart failure symptom improvement, hospitalization and survival.
“LivaNova is dedicated to changing lives through the development and introduction of novel technologies, which address large unmet needs in the global medical community. Autonomic Regulation Therapy is based on our experience derived from VNS Therapy, which is used to treat other disorders,” said Edward Andrle, LivaNova general manager of Neuromodulation. “The ANTHEM-HFrEF Pivotal Study brings us closer to offering the VITARIA System to a large segment of the more than 20 million people currently battling heart failure around the world.”
“Autonomic Regulation Therapy holds promise for patients with advanced heart failure, and we are pleased that this landmark study is underway,” said Dr. Marvin Konstam, principal investigator for the ANTHEM-HFrEF Pivotal Study and chief physician of the CardioVascular Center at Tufts Medical Center in Boston, Mass.
LivaNova PLC is a global medical technology company built on nearly five decades of experience and a relentless commitment to improve the lives of patients around the world. LivaNova’s advanced technologies and breakthrough treatments provide meaningful solutions for the benefit of patients, healthcare professionals and healthcare systems. Headquartered in London, LivaNova has a presence in more than 100 countries worldwide. The company currently employs approximately 4,000 employees. LivaNova operates as two businesses: Cardiac Surgery and Neuromodulation, with operating headquarters in Mirandola, Italy, and Houston, Texas, respectively.