Business Wire10.04.18
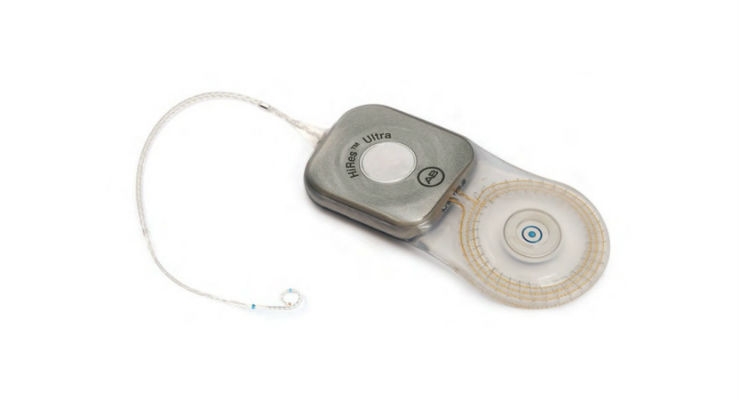
Advanced Bionics (AB) has received U.S. Food and Drug Administration approval of the HiRes Ultra 3D Cochlear Implant. Built on the HiRes Ultra platform and developed by the internal research and development team at AB, the new implant does not require magnet removal or head bandaging, meaning no hearing downtime for the patient.
Hansjuerg Emch, group vice president of Cochlear Implants (CI) at Sonova said, “After many years of research and development, the new magnet technology in our cochlear implants will improve the quality of life for our many recipients due to our hassle free and pain free magnet. We are leading the way and ensuring our recipients have the best possible experiences with our advanced technology. It’s a powerful innovation and makes our efforts so rewarding for patients.”
The new magnet design provides alignment with an external magnetic field in any direction. This allows cochlear implant recipients to move freely around in the strong magnetic field of a magnetic resonance imaging (MRI) machine without feeling pain or discomfort, and without restrictions to the orientation of the head.
MRI examinations are already a standard of care for healthcare professionals and with this in mind AB developed new magnet technology that is compatible with MRI screenings, allowing patients’ peace of mind for any future health issues that necessitate an MRI examination. Previously, patients and surgeons had to contend with the strong magnetic field from MRI machines exerting force on the magnet, causing torque and subsequent pain if the magnet remained in situ, even with head bandaging. Therefore it was common to remove the magnet for high resolution MRI examinations, requiring out-patient surgery and interrupting the patient’s hearing during the healing process. And with the future in mind the new technology has been designed to undergo heavy usage of MRI procedures without any loss of magnetic strength or mobility of the magnet components.
Advanced Bionics develops hearing solutions for individuals with severe-to-profound hearing loss who no longer benefit from hearing aids. Founded in 1993 and a subsidiary of the Sonova Group since 2009, AB develops cochlear implant technology that allows recipients to hear their best.
AB offers the most capable cochlear implant system on the market,1 the HiResolution Bionic Ear System designed to help recipients hear in noisy settings and enjoy the full dimensions of music and tonal languages.2,3
References
1. Technical Specifications. HiRes™ Ultra 3D Cochlear Implant. 2018. 028-M960-03 RevB.
2. Adams D, Ajimsha KM, Barberá MT, Gazibegovic D, Gisbert J, Gómez J, Raveh E, Rocca C, Romanet P, Seebens Y, Zarowski A., Multicentre evaluation of music perception in adult users of Advanced Bionics cochlear implants Cochlear Implants Int. 2014 Jan;15(1):20-6. doi: 10.1179/1754762813Y.0000000032. Epub 2013 Nov 25.
3. Chang YT, Yang HM, Lin YH, Liu SH, Wu JL. Tone discrimination and speech perception benefit in Mandarin-speaking children fit with HiRes Fidelity 120 sound processing. Otol Neurotol. 2009 Sep;30(6):750-7. doi: 10.1097/MAO.0b013e3181b286b2.
Hansjuerg Emch, group vice president of Cochlear Implants (CI) at Sonova said, “After many years of research and development, the new magnet technology in our cochlear implants will improve the quality of life for our many recipients due to our hassle free and pain free magnet. We are leading the way and ensuring our recipients have the best possible experiences with our advanced technology. It’s a powerful innovation and makes our efforts so rewarding for patients.”
The new magnet design provides alignment with an external magnetic field in any direction. This allows cochlear implant recipients to move freely around in the strong magnetic field of a magnetic resonance imaging (MRI) machine without feeling pain or discomfort, and without restrictions to the orientation of the head.
MRI examinations are already a standard of care for healthcare professionals and with this in mind AB developed new magnet technology that is compatible with MRI screenings, allowing patients’ peace of mind for any future health issues that necessitate an MRI examination. Previously, patients and surgeons had to contend with the strong magnetic field from MRI machines exerting force on the magnet, causing torque and subsequent pain if the magnet remained in situ, even with head bandaging. Therefore it was common to remove the magnet for high resolution MRI examinations, requiring out-patient surgery and interrupting the patient’s hearing during the healing process. And with the future in mind the new technology has been designed to undergo heavy usage of MRI procedures without any loss of magnetic strength or mobility of the magnet components.
Advanced Bionics develops hearing solutions for individuals with severe-to-profound hearing loss who no longer benefit from hearing aids. Founded in 1993 and a subsidiary of the Sonova Group since 2009, AB develops cochlear implant technology that allows recipients to hear their best.
AB offers the most capable cochlear implant system on the market,1 the HiResolution Bionic Ear System designed to help recipients hear in noisy settings and enjoy the full dimensions of music and tonal languages.2,3
References
1. Technical Specifications. HiRes™ Ultra 3D Cochlear Implant. 2018. 028-M960-03 RevB.
2. Adams D, Ajimsha KM, Barberá MT, Gazibegovic D, Gisbert J, Gómez J, Raveh E, Rocca C, Romanet P, Seebens Y, Zarowski A., Multicentre evaluation of music perception in adult users of Advanced Bionics cochlear implants Cochlear Implants Int. 2014 Jan;15(1):20-6. doi: 10.1179/1754762813Y.0000000032. Epub 2013 Nov 25.
3. Chang YT, Yang HM, Lin YH, Liu SH, Wu JL. Tone discrimination and speech perception benefit in Mandarin-speaking children fit with HiRes Fidelity 120 sound processing. Otol Neurotol. 2009 Sep;30(6):750-7. doi: 10.1097/MAO.0b013e3181b286b2.