Owlstone Medical08.28.18
Owlstone Medical, a diagnostics company developing a breathalyzer for disease, has expanded its Scientific Advisory Board (SAB) with the appointment of five experts in a range of high-value disciplines.
Owlstone Medical has been advised by Prof. Sir Bruce Ponder, Emeritus Professor of Oncology at the University of Cambridge, since June 2017. In this time, Ponder has played an important role supporting the application of Owlstone Medical’s Breath Biopsy platform to solve some of the most challenging questions in oncology, in particular screening for the early detection of cancer when treatment can be most effective. was the inaugural director of the Cancer Research UK Cambridge Institute (CRUKCI). He became a Fellow of the Royal Society (FRS) in 2001, was knighted for Services to Medicine in 2008, and was awarded the Cancer Research UK Lifetime Achievement Prize in 2013. Ponder has made enormous contributions to the field of oncology, including setting up one of the first specialist clinics for families with cancer, involvement in the discovery of several genes predisposing to cancer, and advances in our understanding of how cancer develops. His group at the CRUKCI is investigating how variations in multiple genes combine to contribute to susceptibility to breast and lung cancer, with the aim to identify high risk groups for targeted cancer screening and prevention.
Ponder is now being joined by experts in areas critical to the development of the business: oncology, personalized medicine, breath testing, biomarker discovery, mass spectrometry, and toxicology and pharmacology. Owlstone Medical’s SAB will provide support and guide the company on product development, technology platform evolution, and clinical program design during its next phase of growth.
Dr. Patrick Bossuyt
Bossuyt is professor of clinical epidemiology at the University of Amsterdam and is chair of the Division of Public Health & Clinical Methods in the Academic Medical Center. He leads the Biomarker and Test Evaluation Research program (BiTE) in Amsterdam, which aims to appraise and develop methods for evaluating medical tests and biomarkers, and to apply these methods in relevant clinical studies. His scientific work spans a broad range of topics in clinical research, developed in close collaboration with clinical departments in the in the Academic Medical Center, with an emphasis on studies of the effectiveness of clinical interventions to guide clinical recommendations and coverage decisions.
The BiTE Program aims to appraise and develop methods for evaluating medical tests and biomarkers, and to apply these methods in relevant clinical studies. In doing so, the program wants to strengthen the evidence-base for rational decision-making about the use of tests and test strategies in healthcare. Bossuyt spearheaded the STARD initiative for the improved reporting of diagnostic test accuracy studies. Bossuyt has authored and co-authored several hundred publications in peer-reviewed journals and serves on the editorial board of a number of these, including Radiology and Clinical Chemistry. He is a member of several national and international advisory committees. He chairs the Scientific Advisory Committee of the Dutch Health Insurance Board, overseeing the national healthcare benefits package.
Dr. Christian Frezza
Frezza is an MRC Programme Leader at the MRC Cancer Unit, University of Cambridge. His research is focused on understanding the role of altered metabolism in cancer, particularly investigating how small molecule metabolites affect the process of tumorigenesis with the goal of to exploit this knowledge to pioneer novel tools for cancer diagnosis and therapy. He studied Medicinal Chemistry at the University of Padova, Italy, and gained his MSc in 2002, after a period of research on mitochondrial toxicity induced by photoactivable anticancer drugs.
Frezza then joined the laboratory of Luca Scorrano in Padova to start a doctorate on mitochondrial dynamics and apoptosis. In 2008, he moved to the Beatson Institute of Cancer Research in Glasgow as recipient of an EMBO Long Term Fellowship, where he investigated the role of mitochondrial defects in tumorigenesis. He moved to the MRC Cancer Unit in 2012, to take up his current position. His research is focused on understanding the role of altered metabolism in cancer, particularly investigating how small molecule metabolites affect the process of tumorigenesis. The major goal of his team is to exploit this knowledge to pioneer novel tools for cancer diagnosis and therapy.
Dr. Chris A. Mayhew
Mayhew is the director of the Institute for Breath Research at the University of Innsbruck and leads the Molecular Physics Group at the school of Physics and Astronomy at the University of Birmingham. His research involves the use of soft chemical ionization mass spectrometric techniques in the fields of health science, atmospheric chemistry and homeland security.
Mayhew studied at the Imperial College of Science and Technology, where he graduated with distinction in 1981 in theoretical physics, and acquired his Ph.D. in molecular physics four years later. He continued his scientific work in the Chemistry Division of the Argonne National Laboratory in Chicago, Ill., where his interest in mass spectrometry was aroused. In 1987 Mayhew became Alexander von Humboldt fellow to the Physical Institute of the University of Bonn, where he was able to perform X-ray molecular mass spectrometric studies thanks to a special synchrotron light resource available in Bonn. During his stay in Germany, Mayhew took a permanent position at the school of Physics and Astronomy at the University of Birmingham (United Kingdom), where he has led the Molecular Physics Group for the next 29 years.
Simultaneously, he created an internationally unique infrastructure in terms of instrumentation to carry out applied and basic research on ion molecule processes. Mayhew's multidisciplinary research involves the use of soft chemical ionization mass spectrometric techniques in the fields of health science, atmospheric chemistry and homeland security. On Oct. 1, 2016, he was appointed director of the Institute for Breath Research at the Leopold-Franzens-Universität Innsbruck, Austria, and on May 1, 2018, he was appointed professor of molecular physics at the University of Birmingham. He currently divides his time between Innsbruck and Birmingham.
Dr. Anil Modak
Formerly, associate director of Medical Products Research & Development at Cambridge Isotope Laboratories, Modak is an expert in stable isotope breath tests and consultant to leading life sciences and technology companies. He has been involved in the design, research, and development of noninvasive breath tests for personalized medicine using stable isotope substrates for the monitoring of disease severity/toxicity and the evaluation of drug metabolizing enzyme activity.
He is the author of several recent patents and publications and numerous presentations at medical conferences around the world. He has reviewed several papers for reputed journals and authored two book chapters. He serves on the editorial board of the Journal of Breath Research, Journal of Pharmacogenomics & Pharmacoproteomics, and International Journal of Clinical Pharmacology & Toxicology. Modak's previous experience includes working for Ribozyme Pharmaceuticals in Boulder, Colo., and Monsanto in St Louis, Mo. His postdoctoral research was conducted at the University of Iowa and Kings College in London.
Dr. Alan Boobis
Boobis is professor of toxicology at Imperial College London. He retired from his position at the College as professor of biochemical pharmacology and director of the Public Health England/ Department of Health-supported Toxicology Unit in June 2017, after over 40 years. His main research interests lie in mechanistic toxicology, drug metabolism, mode of action and chemical risk assessment.
Boobis' main research interests lie in mechanistic toxicology, drug metabolism, mode of action and chemical risk assessment. He has published around 250 original research papers (H-factor 80). He has been a member of several national and international advisory committees; he is currently a member of the U.K. Committee on Toxicity (chair), the WHO Study Group on Tobacco Product Regulation (TobReg), FAO/WHO JECFA (veterinary residues – previous chair) and FAO/WHO JMPR (pesticide residues - previous chair), amongst others. Boobis is a member and a past chairman of the Board of Trustees of ILSI (International Life Sciences Institute) HESI, past president of ILSI Europe and past chair of ILSI. He is involved in several HESI and ILSI Europe projects. Boobis is a fellow of several learned societies and has received a number of awards recognizing his contributions to toxicological sciences, including the civilian award of Officer of the British Empire (OBE).
Billy Boyle, co-founder and CEO at Owlstone Medical, commented, “Owlstone Medical is in a period of rapid growth, with an increasing number of pharmaceutical companies partnering with us to identify novel breath biomarkers; several high profile clinical trials underway in the areas of lung, colorectal, and other forms of cancer; and multiple products in development targeted to reach market in the next twelve months. Supported by recent financing activities, we are set to accelerate our development, and so have established an advisory network to help guide us through our next phase of growth. The SAB forms a key strategic resource for the company and I am very pleased that we have been able to recruit such experienced and well-respected members to join us on our journey.”
Owlstone Medical has developed a unique breathalyzer for disease. Its Breath Biopsy platform is creating a new industry category, based on the routine detection and analysis of volatile organic compound (VOC) biomarkers in breath, which has the potential to revolutionize healthcare. The ReCIVA Breath Sampler is the first standardized breath collection device designed to capture the VOC biomarkers present in breath, which are then analyzed using Owlstone Medical’s Breath Biopsy services and products.
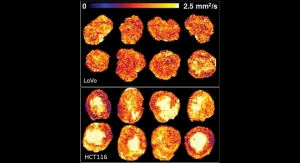
Owlstone Medical is developing breath tests for the early detection of lung and colorectal cancer. These are two of the most common cancer killers worldwide and represent multibillion dollar market opportunities. The company has an active clinical diagnostics/screening pipeline including the world’s largest breath-based clinical trials. The Breath Biopsy platform is also being deployed in the PAN cancer trial, a collaboration with Cancer Research UK studying the early detection of eight different cancer types in breath.
Owlstone Medical was spun out of Owlstone Inc. in 2016 to develop and commercialize medical applications of its proprietary microchip chemical sensing technology, Field Asymmetric Ion Mobility Spectrometry (FAIMS), which is sold in the military and industrial sectors globally. The company has raised a total of $38.5 million in equity financing, and is backed by technology investor Horizons Ventures, and Aviva, a leading healthcare insurer. The company is headquartered in Cambridge, United Kingdom.
Owlstone Medical has been advised by Prof. Sir Bruce Ponder, Emeritus Professor of Oncology at the University of Cambridge, since June 2017. In this time, Ponder has played an important role supporting the application of Owlstone Medical’s Breath Biopsy platform to solve some of the most challenging questions in oncology, in particular screening for the early detection of cancer when treatment can be most effective. was the inaugural director of the Cancer Research UK Cambridge Institute (CRUKCI). He became a Fellow of the Royal Society (FRS) in 2001, was knighted for Services to Medicine in 2008, and was awarded the Cancer Research UK Lifetime Achievement Prize in 2013. Ponder has made enormous contributions to the field of oncology, including setting up one of the first specialist clinics for families with cancer, involvement in the discovery of several genes predisposing to cancer, and advances in our understanding of how cancer develops. His group at the CRUKCI is investigating how variations in multiple genes combine to contribute to susceptibility to breast and lung cancer, with the aim to identify high risk groups for targeted cancer screening and prevention.
Ponder is now being joined by experts in areas critical to the development of the business: oncology, personalized medicine, breath testing, biomarker discovery, mass spectrometry, and toxicology and pharmacology. Owlstone Medical’s SAB will provide support and guide the company on product development, technology platform evolution, and clinical program design during its next phase of growth.
Dr. Patrick Bossuyt
Bossuyt is professor of clinical epidemiology at the University of Amsterdam and is chair of the Division of Public Health & Clinical Methods in the Academic Medical Center. He leads the Biomarker and Test Evaluation Research program (BiTE) in Amsterdam, which aims to appraise and develop methods for evaluating medical tests and biomarkers, and to apply these methods in relevant clinical studies. His scientific work spans a broad range of topics in clinical research, developed in close collaboration with clinical departments in the in the Academic Medical Center, with an emphasis on studies of the effectiveness of clinical interventions to guide clinical recommendations and coverage decisions.
The BiTE Program aims to appraise and develop methods for evaluating medical tests and biomarkers, and to apply these methods in relevant clinical studies. In doing so, the program wants to strengthen the evidence-base for rational decision-making about the use of tests and test strategies in healthcare. Bossuyt spearheaded the STARD initiative for the improved reporting of diagnostic test accuracy studies. Bossuyt has authored and co-authored several hundred publications in peer-reviewed journals and serves on the editorial board of a number of these, including Radiology and Clinical Chemistry. He is a member of several national and international advisory committees. He chairs the Scientific Advisory Committee of the Dutch Health Insurance Board, overseeing the national healthcare benefits package.
Dr. Christian Frezza
Frezza is an MRC Programme Leader at the MRC Cancer Unit, University of Cambridge. His research is focused on understanding the role of altered metabolism in cancer, particularly investigating how small molecule metabolites affect the process of tumorigenesis with the goal of to exploit this knowledge to pioneer novel tools for cancer diagnosis and therapy. He studied Medicinal Chemistry at the University of Padova, Italy, and gained his MSc in 2002, after a period of research on mitochondrial toxicity induced by photoactivable anticancer drugs.
Frezza then joined the laboratory of Luca Scorrano in Padova to start a doctorate on mitochondrial dynamics and apoptosis. In 2008, he moved to the Beatson Institute of Cancer Research in Glasgow as recipient of an EMBO Long Term Fellowship, where he investigated the role of mitochondrial defects in tumorigenesis. He moved to the MRC Cancer Unit in 2012, to take up his current position. His research is focused on understanding the role of altered metabolism in cancer, particularly investigating how small molecule metabolites affect the process of tumorigenesis. The major goal of his team is to exploit this knowledge to pioneer novel tools for cancer diagnosis and therapy.
Dr. Chris A. Mayhew
Mayhew is the director of the Institute for Breath Research at the University of Innsbruck and leads the Molecular Physics Group at the school of Physics and Astronomy at the University of Birmingham. His research involves the use of soft chemical ionization mass spectrometric techniques in the fields of health science, atmospheric chemistry and homeland security.
Mayhew studied at the Imperial College of Science and Technology, where he graduated with distinction in 1981 in theoretical physics, and acquired his Ph.D. in molecular physics four years later. He continued his scientific work in the Chemistry Division of the Argonne National Laboratory in Chicago, Ill., where his interest in mass spectrometry was aroused. In 1987 Mayhew became Alexander von Humboldt fellow to the Physical Institute of the University of Bonn, where he was able to perform X-ray molecular mass spectrometric studies thanks to a special synchrotron light resource available in Bonn. During his stay in Germany, Mayhew took a permanent position at the school of Physics and Astronomy at the University of Birmingham (United Kingdom), where he has led the Molecular Physics Group for the next 29 years.
Simultaneously, he created an internationally unique infrastructure in terms of instrumentation to carry out applied and basic research on ion molecule processes. Mayhew's multidisciplinary research involves the use of soft chemical ionization mass spectrometric techniques in the fields of health science, atmospheric chemistry and homeland security. On Oct. 1, 2016, he was appointed director of the Institute for Breath Research at the Leopold-Franzens-Universität Innsbruck, Austria, and on May 1, 2018, he was appointed professor of molecular physics at the University of Birmingham. He currently divides his time between Innsbruck and Birmingham.
Dr. Anil Modak
Formerly, associate director of Medical Products Research & Development at Cambridge Isotope Laboratories, Modak is an expert in stable isotope breath tests and consultant to leading life sciences and technology companies. He has been involved in the design, research, and development of noninvasive breath tests for personalized medicine using stable isotope substrates for the monitoring of disease severity/toxicity and the evaluation of drug metabolizing enzyme activity.
He is the author of several recent patents and publications and numerous presentations at medical conferences around the world. He has reviewed several papers for reputed journals and authored two book chapters. He serves on the editorial board of the Journal of Breath Research, Journal of Pharmacogenomics & Pharmacoproteomics, and International Journal of Clinical Pharmacology & Toxicology. Modak's previous experience includes working for Ribozyme Pharmaceuticals in Boulder, Colo., and Monsanto in St Louis, Mo. His postdoctoral research was conducted at the University of Iowa and Kings College in London.
Dr. Alan Boobis
Boobis is professor of toxicology at Imperial College London. He retired from his position at the College as professor of biochemical pharmacology and director of the Public Health England/ Department of Health-supported Toxicology Unit in June 2017, after over 40 years. His main research interests lie in mechanistic toxicology, drug metabolism, mode of action and chemical risk assessment.
Boobis' main research interests lie in mechanistic toxicology, drug metabolism, mode of action and chemical risk assessment. He has published around 250 original research papers (H-factor 80). He has been a member of several national and international advisory committees; he is currently a member of the U.K. Committee on Toxicity (chair), the WHO Study Group on Tobacco Product Regulation (TobReg), FAO/WHO JECFA (veterinary residues – previous chair) and FAO/WHO JMPR (pesticide residues - previous chair), amongst others. Boobis is a member and a past chairman of the Board of Trustees of ILSI (International Life Sciences Institute) HESI, past president of ILSI Europe and past chair of ILSI. He is involved in several HESI and ILSI Europe projects. Boobis is a fellow of several learned societies and has received a number of awards recognizing his contributions to toxicological sciences, including the civilian award of Officer of the British Empire (OBE).
Billy Boyle, co-founder and CEO at Owlstone Medical, commented, “Owlstone Medical is in a period of rapid growth, with an increasing number of pharmaceutical companies partnering with us to identify novel breath biomarkers; several high profile clinical trials underway in the areas of lung, colorectal, and other forms of cancer; and multiple products in development targeted to reach market in the next twelve months. Supported by recent financing activities, we are set to accelerate our development, and so have established an advisory network to help guide us through our next phase of growth. The SAB forms a key strategic resource for the company and I am very pleased that we have been able to recruit such experienced and well-respected members to join us on our journey.”
Owlstone Medical has developed a unique breathalyzer for disease. Its Breath Biopsy platform is creating a new industry category, based on the routine detection and analysis of volatile organic compound (VOC) biomarkers in breath, which has the potential to revolutionize healthcare. The ReCIVA Breath Sampler is the first standardized breath collection device designed to capture the VOC biomarkers present in breath, which are then analyzed using Owlstone Medical’s Breath Biopsy services and products.
Owlstone Medical is developing breath tests for the early detection of lung and colorectal cancer. These are two of the most common cancer killers worldwide and represent multibillion dollar market opportunities. The company has an active clinical diagnostics/screening pipeline including the world’s largest breath-based clinical trials. The Breath Biopsy platform is also being deployed in the PAN cancer trial, a collaboration with Cancer Research UK studying the early detection of eight different cancer types in breath.
Owlstone Medical was spun out of Owlstone Inc. in 2016 to develop and commercialize medical applications of its proprietary microchip chemical sensing technology, Field Asymmetric Ion Mobility Spectrometry (FAIMS), which is sold in the military and industrial sectors globally. The company has raised a total of $38.5 million in equity financing, and is backed by technology investor Horizons Ventures, and Aviva, a leading healthcare insurer. The company is headquartered in Cambridge, United Kingdom.