GlobeNewswire04.12.18
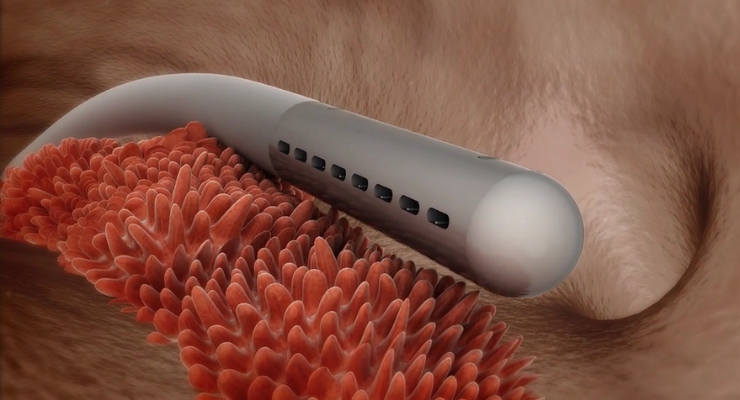
Microbot Medical Inc., a medical device company specializing in the design and development of transformational micro-robotic medical technologies, today announced the European Commission, recognized the company’s innovative Self-Cleaning Shunt (SCS) product and awarded the company a non-dilutive grant to continue developing the SCS. The Commission’s decision, in part, is based upon substantial demand for the SCS with the potential to create new market opportunities.
“The acknowledgment by the European Commission and awarding of the grant is yet another vote of confidence in the continued development process of our unique technology, size of the unmet medical need that we are solving and the value proposition of our SCS product,” commented Harel Gadot, Microbot’s CEO, president, and Chairman. “We continue focusing on our primary objectives to build shareholder value, one of which is to strengthen our balance sheet through non-dilutive sources, to ensure we continue our development and commercialization plans flawlessly. This achievement, coupled with additional milestones we are expecting in the near term such as closing the CardioSert acquisition and completing the two ongoing studies related to our lead SCS product, will ensure momentum over the foreseeable future.”
The current approved 50,000€ grant which the company can submit an additional request in six months for a higher grant up to 2,000,000€, will be used for the continued development of the company’s Self Cleaning Shunt (SCS) product. The company believes that it will reach its anticipated development milestones for its SCS, including its submission with the US Food and Drug Administration (FDA) in late 2018 or early 2019.
“The acknowledgment by the European Commission and awarding of the grant is yet another vote of confidence in the continued development process of our unique technology, size of the unmet medical need that we are solving and the value proposition of our SCS product,” commented Harel Gadot, Microbot’s CEO, president, and Chairman. “We continue focusing on our primary objectives to build shareholder value, one of which is to strengthen our balance sheet through non-dilutive sources, to ensure we continue our development and commercialization plans flawlessly. This achievement, coupled with additional milestones we are expecting in the near term such as closing the CardioSert acquisition and completing the two ongoing studies related to our lead SCS product, will ensure momentum over the foreseeable future.”
The current approved 50,000€ grant which the company can submit an additional request in six months for a higher grant up to 2,000,000€, will be used for the continued development of the company’s Self Cleaning Shunt (SCS) product. The company believes that it will reach its anticipated development milestones for its SCS, including its submission with the US Food and Drug Administration (FDA) in late 2018 or early 2019.