Micro Interventional Devices Inc.06.14.16
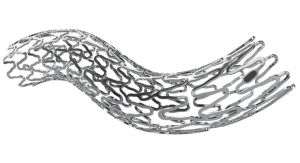
Micro Interventional Devices Inc. (MID), a provider of minimally invasive and catheter-based compliant fixation technology, has received CE Mark approval for the company’s first product: the Permaseal transapical access and closure device. The Permaseal device allows surgeons to access and close the left-ventricle instantaneously, reliably and without suturing the myocardium. Permaseal is being touted as the world’s first device using compliant soft-tissue PolyCorTM anchor technology.
Permaseal was validated in the STASIS Clinical Study (Secure Transapical Access and Closure Study) conducted at five European sites. Results indicated that Permaseal shortened operating time and hospital stay, reduced adverse events including the need for transfusion and reduced 12-month mortality and stroke rates to 0 percent.* The technology provides a direct, safe and simple, access and closure site for emerging complex structural heart disease procedures including TAVR, TMVR, PFO, and other minimally invasive cardiac procedures.
Watch the video below to learn more about the Permaseal device:
“The CE Mark approval of Permaseal is a tremendous achievement for MID,” said Michael Whitman, MID’s founder, president and CEO. “Permaseal is the first in a series of products designed to replace the need for suturing in structural heart procedures. We are excited that this technology is now available to our European surgeons and their patients.”
“In the near future, PolyCor and MyoLast technologies will be utilized in a broad range of proprietary, catheter-based products, addressing unmet needs in tricuspid repair, mitral repair and mitral valve fixation,” added Whitman. “MID’s technology platform enables open-surgical procedures to be performed percutaneously.”
Permaseal is currently the only transapical access and closure device available for commercial use in the European Union. U.S. Food and Drug Administration clearance to market Permaseal is pending.
MID develops compliant fixation technology addressing unmet needs in structural heart disease. The company is based in Newton, Pa.
*Data on file.
Permaseal was validated in the STASIS Clinical Study (Secure Transapical Access and Closure Study) conducted at five European sites. Results indicated that Permaseal shortened operating time and hospital stay, reduced adverse events including the need for transfusion and reduced 12-month mortality and stroke rates to 0 percent.* The technology provides a direct, safe and simple, access and closure site for emerging complex structural heart disease procedures including TAVR, TMVR, PFO, and other minimally invasive cardiac procedures.
Watch the video below to learn more about the Permaseal device:
“The CE Mark approval of Permaseal is a tremendous achievement for MID,” said Michael Whitman, MID’s founder, president and CEO. “Permaseal is the first in a series of products designed to replace the need for suturing in structural heart procedures. We are excited that this technology is now available to our European surgeons and their patients.”
“In the near future, PolyCor and MyoLast technologies will be utilized in a broad range of proprietary, catheter-based products, addressing unmet needs in tricuspid repair, mitral repair and mitral valve fixation,” added Whitman. “MID’s technology platform enables open-surgical procedures to be performed percutaneously.”
Permaseal is currently the only transapical access and closure device available for commercial use in the European Union. U.S. Food and Drug Administration clearance to market Permaseal is pending.
MID develops compliant fixation technology addressing unmet needs in structural heart disease. The company is based in Newton, Pa.
*Data on file.