10.22.15
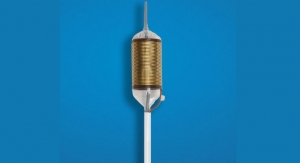
Redwood City, Calif.-based C2 Therapeutics Inc. has earned the CE mark for is Coldplay Cryoballoon focal ablation system. The product is available for commercial use initially in parts of Western Europe. Cryoablation is a process that uses extreme cold to precisely destroy or damage diseased tissue while minimizing damage to surrounding healthy tissue.
“C2’s Coldplay Cryoballoon technology blends the precision and controlled depth of radiofrequency ablation with the inherent benefits of cryotherapy, which until recently was lacking in control and visibility during therapy delivery,” said Jacques Bergman, M.D., Ph.D., professor of gastrointestinal endoscopy, director of endoscopy, Academic Medical Center, Amsterdam, The Netherlands, and co-principal investigator of the Coldplay clinical study. “Coldplay’s unique characteristics include its contained cryotherapy diffuser—housed inside a self-sizing balloon—and an ability to direct cryoablation to targeted tissue in various zones. The CryoBalloon offers a solution that has the potential to address unmet medical needs in treating early Barrett's neoplasia.”
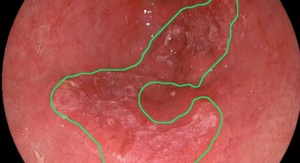
Barrett’s esophagus develops as a result of chronic injury from gastroesophageal reflux disease (GERD). Over time, the normal esophageal lining is replaced with abnormal cells (Barrett’s Esophagus), putting patients at greater risk of developing cancer of the esophagus.
“Gaining the CE mark for the Coldplay system is an important milestone for C2 Therapeutics and sets the stage for the next phase of our corporate evolution as we develop new products to treat patients with Barrett’s Esophagus and other conditions that can be targeted with our novel cryoablation technology,” said Peter Garcia-Meza, president and CEO of C2 Therapeutics. “CryoBalloon Focal offers several advantages over currently available options including reduced treatment duration, decreased post-procedure pain and broad utility in patients who may not be suitable candidates for radiofrequency ablation.”
C2 Therapeutics was founded in 2007 to address the limitations of current Barrett’s esophagus treatment options.
“C2’s Coldplay Cryoballoon technology blends the precision and controlled depth of radiofrequency ablation with the inherent benefits of cryotherapy, which until recently was lacking in control and visibility during therapy delivery,” said Jacques Bergman, M.D., Ph.D., professor of gastrointestinal endoscopy, director of endoscopy, Academic Medical Center, Amsterdam, The Netherlands, and co-principal investigator of the Coldplay clinical study. “Coldplay’s unique characteristics include its contained cryotherapy diffuser—housed inside a self-sizing balloon—and an ability to direct cryoablation to targeted tissue in various zones. The CryoBalloon offers a solution that has the potential to address unmet medical needs in treating early Barrett's neoplasia.”
Barrett’s esophagus develops as a result of chronic injury from gastroesophageal reflux disease (GERD). Over time, the normal esophageal lining is replaced with abnormal cells (Barrett’s Esophagus), putting patients at greater risk of developing cancer of the esophagus.
“Gaining the CE mark for the Coldplay system is an important milestone for C2 Therapeutics and sets the stage for the next phase of our corporate evolution as we develop new products to treat patients with Barrett’s Esophagus and other conditions that can be targeted with our novel cryoablation technology,” said Peter Garcia-Meza, president and CEO of C2 Therapeutics. “CryoBalloon Focal offers several advantages over currently available options including reduced treatment duration, decreased post-procedure pain and broad utility in patients who may not be suitable candidates for radiofrequency ablation.”
C2 Therapeutics was founded in 2007 to address the limitations of current Barrett’s esophagus treatment options.