08.13.15
Tustin, Calif.-based Uptake Medical Corporation has received CE mark approval for its Intervapor system in Europe. Intervapor uses heated water vapor to achieve bronchoscopic lung volume reduction in patients with heterogeneous upper lobe emphysema. According to The Organisation for Economic Co-operation and Development, more than 23 million people in Europe suffer from emphysema, a debilitating and progressive condition that affects the respiratory system.
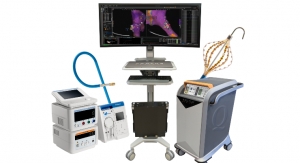
The Intervapor system is comprised of three components: the Intervapor Personalized Procedure Program (IP3), Intervapor catheter, and Intervapor generator. The IP3 recommends diseased lung areas to be treated by precise, physician-controlled doses of thermal energy created by the generator and delivered by the catheter (in the form of heated water vapor) through a standard bronchoscope.
“The traditional treatment options for severe emphysema patients have been very limited which is why we have remained committed over the past decade to finding a more meaningful treatment alternative for this progressive disease,” said King Nelson, president and CEO of Uptake Medical. “Gaining CE mark is a major milestone for our company, but more importantly, represents new hope for emphysema patients. We are eager to begin the commercial launch of Intervapor in Europe allowing more interventional pulmonologists to begin using the system in the months ahead.”
The CE Mark follows results of the Step-Up study, a prospective, multi-center, multi-national, randomized-controlled study that was completed in late 2014. The trial, which involved patients from 13 centers in Europe and Australia, was designed to evaluate the safety and performance of the Intervapor system in addition to the optimal medical standard therapy in improving lung performance and quality of life of patients with severe emphysema. The Step-Up trial results will be reported at the European Respiratory Society meeting in Amsterdam, The Netherlands, in late September 2015.
“Being granted the CE mark validates for Uptake Medical the merits of our clinical hypothesis to have for the first time an option where we can treat the most diseased segments and preserve the less diseased segments,” said Felix Herth, M.D., Step-Up’s co-principal investigator from ThoraxKlinik in Heidelberg, Germany.
Uptake Medical makes non-surgical technology for the treatment of lung cancer and emphysema.
The Intervapor system is comprised of three components: the Intervapor Personalized Procedure Program (IP3), Intervapor catheter, and Intervapor generator. The IP3 recommends diseased lung areas to be treated by precise, physician-controlled doses of thermal energy created by the generator and delivered by the catheter (in the form of heated water vapor) through a standard bronchoscope.
“The traditional treatment options for severe emphysema patients have been very limited which is why we have remained committed over the past decade to finding a more meaningful treatment alternative for this progressive disease,” said King Nelson, president and CEO of Uptake Medical. “Gaining CE mark is a major milestone for our company, but more importantly, represents new hope for emphysema patients. We are eager to begin the commercial launch of Intervapor in Europe allowing more interventional pulmonologists to begin using the system in the months ahead.”
The CE Mark follows results of the Step-Up study, a prospective, multi-center, multi-national, randomized-controlled study that was completed in late 2014. The trial, which involved patients from 13 centers in Europe and Australia, was designed to evaluate the safety and performance of the Intervapor system in addition to the optimal medical standard therapy in improving lung performance and quality of life of patients with severe emphysema. The Step-Up trial results will be reported at the European Respiratory Society meeting in Amsterdam, The Netherlands, in late September 2015.
“Being granted the CE mark validates for Uptake Medical the merits of our clinical hypothesis to have for the first time an option where we can treat the most diseased segments and preserve the less diseased segments,” said Felix Herth, M.D., Step-Up’s co-principal investigator from ThoraxKlinik in Heidelberg, Germany.
Uptake Medical makes non-surgical technology for the treatment of lung cancer and emphysema.