05.15.15
OncoCyte Corporation, subsidiary of BioTime Inc., has received a new round of financing to fund further development and commercialization activities of its cancer diagnostic products. With completion of the financing, which included a cash investment of $3.3 million and conversion of $3.3 million of existing debt into equity, OncoCyte has approximately $7 million in cash and marketable securities, which should be sufficient to fund current operations into early 2016. Proceeds from this transaction will be used to fund the research, clinical development and commercialization of OncoCyte’s proprietary PanC-Dx cancer diagnostic tests. All of OncoCyte’s existing shareholders participated in the financing round.
“With this round of financing, OncoCyte is well positioned to prepare for commercialization of our cancer diagnostic products,” said Joseph Wagner, Ph.D., CEO of OncoCyte. “As we complete analysis of the initial clinical data from our studies in lung, bladder, and breast cancer, we plan on collecting additional clinical validation data for our tests, solidifying the commercialization paths for each product, identifying our lead test for launch, and developing a longer-term financing strategy. On behalf of OncoCyte, I thank our dedicated investors for their continued support.”
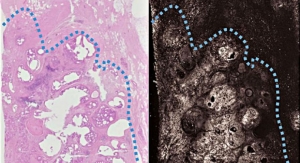
PanC-Dx is a class of non-invasive cancer diagnostics based on OncoCyte’s proprietary set of cancer markers, which were discovered by company scientists through an analysis of broad gene expression patterns in numerous cancer types. OncoCyte is currently sponsoring three clinical studies of PanC-Dx in bladder, breast, and lung cancer.
Based in Alameda, Calif., OncoCyte is developing cancer treatment and diagnosis technology.
“With this round of financing, OncoCyte is well positioned to prepare for commercialization of our cancer diagnostic products,” said Joseph Wagner, Ph.D., CEO of OncoCyte. “As we complete analysis of the initial clinical data from our studies in lung, bladder, and breast cancer, we plan on collecting additional clinical validation data for our tests, solidifying the commercialization paths for each product, identifying our lead test for launch, and developing a longer-term financing strategy. On behalf of OncoCyte, I thank our dedicated investors for their continued support.”
PanC-Dx is a class of non-invasive cancer diagnostics based on OncoCyte’s proprietary set of cancer markers, which were discovered by company scientists through an analysis of broad gene expression patterns in numerous cancer types. OncoCyte is currently sponsoring three clinical studies of PanC-Dx in bladder, breast, and lung cancer.
Based in Alameda, Calif., OncoCyte is developing cancer treatment and diagnosis technology.