05.14.15
Wayne, Pa.-based Intact Vascular Inc., which develops minimally invasive peripheral vascular procedures, has raised $38.9 million in a series B venture capital financing. The funds will be used to accelerate the development and U.S. Food and Drug Administration approval of the company’s Tack endovascular system. The latest round, one of the largest private medical technology financings of 2015, was led by New Enterprise Associates (NEA), with participation from Quaker Partners, H.I.G. BioVentures and other existing investors.
Related to the financing, NEA Partner Justin Klein, M.D., JD, has joined the company’s board of directors.
“We are poised to build significant value with the conduct of multiple clinical trials, both above and below the knee, all of which are aimed at demonstrating the unique value of the Tack Endovascular System in the treatment of peripheral artery disease and moving us through the FDA approval process,” said Bruce Shook, president and CEO of Intact Vascular. “We are thrilled to have the support of NEA as the lead investor in this Series B round. NEA’s track record of success and the expertise it brings to the table make them an ideal partner for Intact Vascular.”
“Intact Vascular’s approach to peripheral artery disease is truly novel and has the potential to significantly improve treatment of this rapidly growing disease,” said Klein. “We are excited to partner with the outstanding team at Intact Vascular and their investors to move rapidly into pivotal clinical trial work in peripheral artery disease and to explore new applications of this novel approach to vascular repair.”
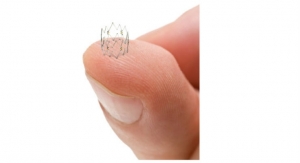
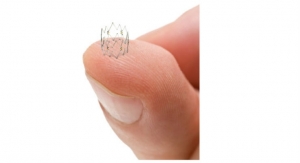
Intact Vascular was founded in 2012 to develop the technology pioneered by Peter Schneider, M.D., director of the Division of Vascular Therapy at the Kaiser Foundation Hospital in Honolulu, Hawaii. Schneider envisioned a new, endovascular method for repairing arterial dissections following percutaneous transluminal angioplasty to permit highly targeted repair of diseased arteries, while minimizing the metal left behind and inflammation of the artery wall, both of which are significant problems associated with stenting, the current method of dissection repair.
Over the last three years, Intact Vascular has developed and refined the Tack endovascular system implant and delivery system, conducted three international clinical trials, and obtained a CE mark for the above-the-knee system. The clinical trials enrolled more than 170 patients and demonstrated the promise of this new approach to arterial dissection repair. Peripheral artery disease impacts more than 200 million people worldwide and more than 19 million people in the U.S. The prevalence of peripheral artery disease among American adults exceeds that of coronary heart disease and its growth is being driven by the triad of obesity, diabetes and advancing age.
Related to the financing, NEA Partner Justin Klein, M.D., JD, has joined the company’s board of directors.
“We are poised to build significant value with the conduct of multiple clinical trials, both above and below the knee, all of which are aimed at demonstrating the unique value of the Tack Endovascular System in the treatment of peripheral artery disease and moving us through the FDA approval process,” said Bruce Shook, president and CEO of Intact Vascular. “We are thrilled to have the support of NEA as the lead investor in this Series B round. NEA’s track record of success and the expertise it brings to the table make them an ideal partner for Intact Vascular.”
“Intact Vascular’s approach to peripheral artery disease is truly novel and has the potential to significantly improve treatment of this rapidly growing disease,” said Klein. “We are excited to partner with the outstanding team at Intact Vascular and their investors to move rapidly into pivotal clinical trial work in peripheral artery disease and to explore new applications of this novel approach to vascular repair.”
Intact Vascular was founded in 2012 to develop the technology pioneered by Peter Schneider, M.D., director of the Division of Vascular Therapy at the Kaiser Foundation Hospital in Honolulu, Hawaii. Schneider envisioned a new, endovascular method for repairing arterial dissections following percutaneous transluminal angioplasty to permit highly targeted repair of diseased arteries, while minimizing the metal left behind and inflammation of the artery wall, both of which are significant problems associated with stenting, the current method of dissection repair.
Over the last three years, Intact Vascular has developed and refined the Tack endovascular system implant and delivery system, conducted three international clinical trials, and obtained a CE mark for the above-the-knee system. The clinical trials enrolled more than 170 patients and demonstrated the promise of this new approach to arterial dissection repair. Peripheral artery disease impacts more than 200 million people worldwide and more than 19 million people in the U.S. The prevalence of peripheral artery disease among American adults exceeds that of coronary heart disease and its growth is being driven by the triad of obesity, diabetes and advancing age.