01.21.15
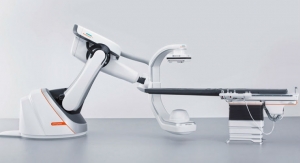
Lake Oswego, Ore.-based Biotronik, a cardiovascular medical technology company, has completed enrollment in phase C of its ProMRI study. This phase investigates the safety of implantable cardioverter defibrillator (ICD) devices in patients undergoing full-body magnetic resonance imaging (MRI) scans. The Biotronik DX system is also included in the study that investigates the effects of cardiac and thoracic spinal scans. Biotronik is one of only two companies in the United States currently approved to conduct these studies, which aim to broaden diagnostic options for cardiac device patients.
The Biotronik-sponsored trial has been in U.S. subject recruitment since March 2013.
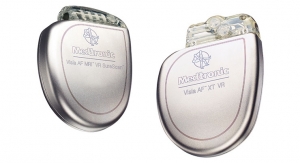
The Biotronik DX ICD system is designed to give the complete diagnostic capabilities of a dual-chamber ICD, including atrial sensing and advanced proven shock reduction algorithms, with only a single lead. DX technology was first introduced in the United States in 2013. According to Biotronik, the DX appeals to patients, who prefer lighter implants, and to physicians, who benefit from dual-chamber diagnostic information.
The Biotronik ProMRI study consists of a series of MRI compatibility trials intended to increase cardiac device patients’ access to MRI. Phase A resulted in FDA approval of Entovis pacemaker systems for MRI scans with an exclusion zone. Phase B results were submitted to the Food and Drug Administration (FDA) for approval of the Entovis and Eluna pacemaker systems for full-body MRI scans. Biotronik plans on submitting data from phase C to the FDA in 2015.
There are more than 6.5 million pacemaker patients worldwide and more than one million are given implants each year. Until recently, pacemaker and ICD patients could not get MRI scans due to concern that the strong magnetic fields and radio waves created by an MRI could have a negative effect on both device and patient. Biotronik’s ProMRI technology allows patients with the implanted pacemaker to receive and MRI.
The Biotronik-sponsored trial has been in U.S. subject recruitment since March 2013.
The Biotronik DX ICD system is designed to give the complete diagnostic capabilities of a dual-chamber ICD, including atrial sensing and advanced proven shock reduction algorithms, with only a single lead. DX technology was first introduced in the United States in 2013. According to Biotronik, the DX appeals to patients, who prefer lighter implants, and to physicians, who benefit from dual-chamber diagnostic information.
The Biotronik ProMRI study consists of a series of MRI compatibility trials intended to increase cardiac device patients’ access to MRI. Phase A resulted in FDA approval of Entovis pacemaker systems for MRI scans with an exclusion zone. Phase B results were submitted to the Food and Drug Administration (FDA) for approval of the Entovis and Eluna pacemaker systems for full-body MRI scans. Biotronik plans on submitting data from phase C to the FDA in 2015.
There are more than 6.5 million pacemaker patients worldwide and more than one million are given implants each year. Until recently, pacemaker and ICD patients could not get MRI scans due to concern that the strong magnetic fields and radio waves created by an MRI could have a negative effect on both device and patient. Biotronik’s ProMRI technology allows patients with the implanted pacemaker to receive and MRI.