12.04.14
Santa Rosa, Calif.-based Direct Flow Medical Inc. closed a new round of financing totaling $32 million.
The company also appointed Chris Richardson as its first chief commercial officer in order to, according to the company, “support global growth” of the Direct Flow Medical Transcatheter Aortic Valve System.
The latest financing round includes $17 million in private equity, led by new investor SV Life Sciences and a new strategic investor, and the second tranche of a $50 million debt facility from PDL BioPharma worth $15 million. Proceeds will be used to complete the U.S. SALUS pivotal trial and help expand the Direct Flow Medical system outside of the U.S., company officials said.
“Direct Flow Medical has developed a true, second-generation transcatheter aortic replacement system that solves many of the clinical issues associated with earlier devices. This platform technology is not only applicable to the treatment of aortic stenosis, but also holds tremendous promise for the treatment of aortic insufficiency and mitral disease,” said Paul LaViolette, Direct Flow Medical chairman and managing director of SV LifeSciences. “SV Life Sciences is pleased to join the existing group of investors at Direct Flow Medical and provide the capital necessary to build the company’s global business in 2015 and beyond.”
Richardson has spent his 25-year career taking cardiac startup companies from the clinical trial phase through worldwide commercialization and into acquisition.
Prior to joining Direct Flow Medical, he was General Manager at Abbott Vascular Structural Heart, a global company focused on percutaneous mitral valve repair, having joined Abbott through its acquisition of Evalve. Before Evalve, he was area vice president for the United States at Boston Scientific in its Cardiac Rhythm Management business. Prior to that, Richardson served as director of GSC Management Development at Guidant, where he was responsible for all senior management leadership programs. Earlier, he held leadership positions at Heartport (acquired by Johnson & Johnson) and Devices for Vascular Intervention (acquired by Guidant). Richardson holds a B.S. in psychology and biology from Indiana University.
“We strive to strengthen our management team at key inflection points in the company’s lifespan, and Chris joins us at a time of rapid growth,” said Bernard E. Lyons, Ph.D., Direct Flow’s president and CEO. “We are excited to add his expertise and experience to our leadership team as we continue to expand our market share in Europe and focus on enrollment for the SALUS pivotal trial in the U.S.”
With the addition of Richardson, Dan Rose has been appointed vice president and general manager of Europe, Middle East and Africa. Based in Europe, Rose will continue to oversee the company’s worldwide marketing efforts in his new role.
The Direct Flow Medical Transcatheter Aortic Valve System is commercially available in Europe. In the United States, the company is progressing toward regulatory approval and currently is enrolling patients in its SALUS pivotal trial.
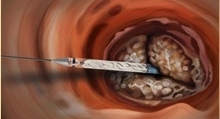
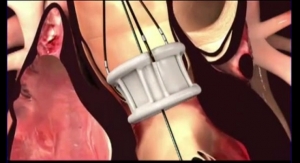
Rather than a metal stent, Direct Flow’s system incorporates a polymer frame, which is expanded using pressurized saline and contrast for placement, assessment and repositioning. The saline/contrast solution is easily exchanged for a quick-curing polymer that solidifies and secures the valve in place once optimal positioning is reached, according to the company. The double-ring design of the valve creates a tight seal around the annulus. The system is fully repositionable and retrievable up until polymer exchange.
The system avoids rapid pacing of the heart during deployment, and does not require post-dilatation following placement. The system is indicated for the treatment of patients with severe aortic stenosis who are at extreme risk for surgical aortic valve replacement. The metal-free design enables a low-profile, fully sheathed delivery system for all valve sizes that minimizes vascular complications and improves hemodynamic outcomes.
In the transcatheter aortic valve replacement (TAVR) market in the United States, Direct Flow is going up against industry heavy-hitters Edwards Lifesciences Corp. and Medtronic Inc., both of which have TAVR systems approved in the United States and have released next-generation iterations for new indications.
Direct Flow Medical was founded in 2004. In addition to its headquarters in Santa Rosa, the company also has technology and manufacturing facilities in Lake Forest, Calif. The company’s investors include EDF Ventures, New Leaf Venture Partners, Spray Venture Partners, Foundation Medical Partners, SV Life Sciences, VantagePoint Venture Partners, ePlanet Venture Partners and strategic corporate investors.
The company also appointed Chris Richardson as its first chief commercial officer in order to, according to the company, “support global growth” of the Direct Flow Medical Transcatheter Aortic Valve System.
The latest financing round includes $17 million in private equity, led by new investor SV Life Sciences and a new strategic investor, and the second tranche of a $50 million debt facility from PDL BioPharma worth $15 million. Proceeds will be used to complete the U.S. SALUS pivotal trial and help expand the Direct Flow Medical system outside of the U.S., company officials said.
“Direct Flow Medical has developed a true, second-generation transcatheter aortic replacement system that solves many of the clinical issues associated with earlier devices. This platform technology is not only applicable to the treatment of aortic stenosis, but also holds tremendous promise for the treatment of aortic insufficiency and mitral disease,” said Paul LaViolette, Direct Flow Medical chairman and managing director of SV LifeSciences. “SV Life Sciences is pleased to join the existing group of investors at Direct Flow Medical and provide the capital necessary to build the company’s global business in 2015 and beyond.”
Richardson has spent his 25-year career taking cardiac startup companies from the clinical trial phase through worldwide commercialization and into acquisition.
Prior to joining Direct Flow Medical, he was General Manager at Abbott Vascular Structural Heart, a global company focused on percutaneous mitral valve repair, having joined Abbott through its acquisition of Evalve. Before Evalve, he was area vice president for the United States at Boston Scientific in its Cardiac Rhythm Management business. Prior to that, Richardson served as director of GSC Management Development at Guidant, where he was responsible for all senior management leadership programs. Earlier, he held leadership positions at Heartport (acquired by Johnson & Johnson) and Devices for Vascular Intervention (acquired by Guidant). Richardson holds a B.S. in psychology and biology from Indiana University.
“We strive to strengthen our management team at key inflection points in the company’s lifespan, and Chris joins us at a time of rapid growth,” said Bernard E. Lyons, Ph.D., Direct Flow’s president and CEO. “We are excited to add his expertise and experience to our leadership team as we continue to expand our market share in Europe and focus on enrollment for the SALUS pivotal trial in the U.S.”
With the addition of Richardson, Dan Rose has been appointed vice president and general manager of Europe, Middle East and Africa. Based in Europe, Rose will continue to oversee the company’s worldwide marketing efforts in his new role.
The Direct Flow Medical Transcatheter Aortic Valve System is commercially available in Europe. In the United States, the company is progressing toward regulatory approval and currently is enrolling patients in its SALUS pivotal trial.
Rather than a metal stent, Direct Flow’s system incorporates a polymer frame, which is expanded using pressurized saline and contrast for placement, assessment and repositioning. The saline/contrast solution is easily exchanged for a quick-curing polymer that solidifies and secures the valve in place once optimal positioning is reached, according to the company. The double-ring design of the valve creates a tight seal around the annulus. The system is fully repositionable and retrievable up until polymer exchange.
The system avoids rapid pacing of the heart during deployment, and does not require post-dilatation following placement. The system is indicated for the treatment of patients with severe aortic stenosis who are at extreme risk for surgical aortic valve replacement. The metal-free design enables a low-profile, fully sheathed delivery system for all valve sizes that minimizes vascular complications and improves hemodynamic outcomes.
In the transcatheter aortic valve replacement (TAVR) market in the United States, Direct Flow is going up against industry heavy-hitters Edwards Lifesciences Corp. and Medtronic Inc., both of which have TAVR systems approved in the United States and have released next-generation iterations for new indications.
Direct Flow Medical was founded in 2004. In addition to its headquarters in Santa Rosa, the company also has technology and manufacturing facilities in Lake Forest, Calif. The company’s investors include EDF Ventures, New Leaf Venture Partners, Spray Venture Partners, Foundation Medical Partners, SV Life Sciences, VantagePoint Venture Partners, ePlanet Venture Partners and strategic corporate investors.